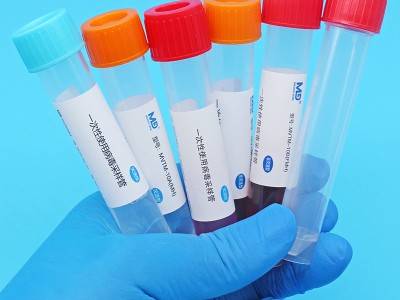
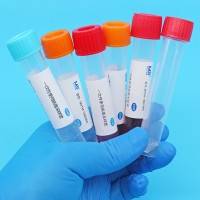
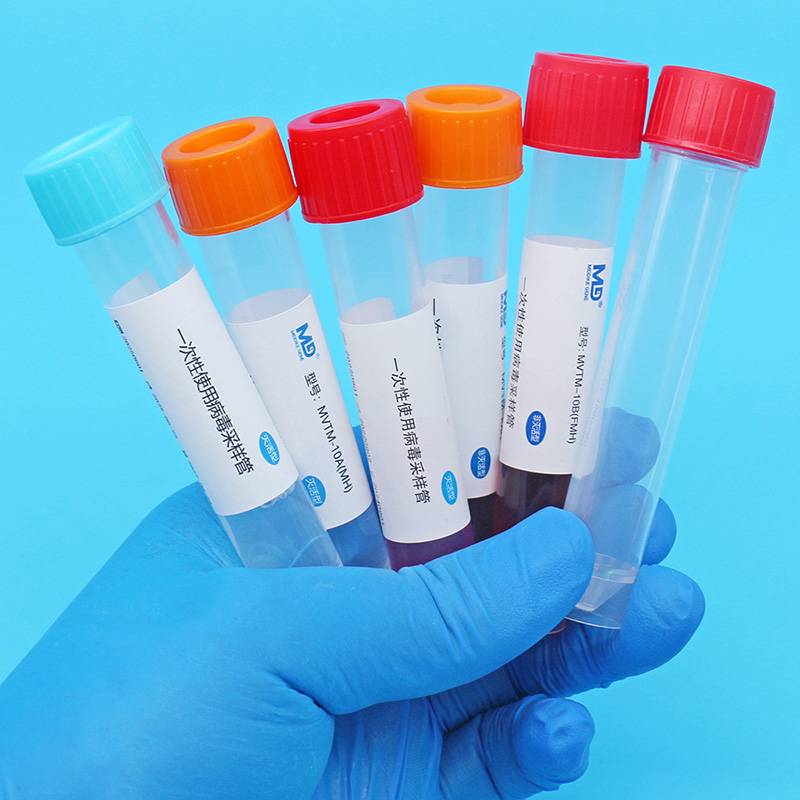
Virus sampling tubes can also be used to transport tissue samples from sampling sites to testing laboratories that swab or specific locations, pending extraction and testing by the pcr and necessary cell culture。
Materials
Medical pp polypropylene
Composition
Seal cap + tube + active saver + label bar code etc。
Specification model
Mvtm-2a (mh), mvtm-5a (mh), mvtm-10a (mh), mvtm-15a (mh), mvtm-30a (mh), mvtm-2b (mh), mvtm-5b (mh), mvtm-10b (mh), mvtm-15b (mh), mvtm-30b (mh)。
A: swab b: swab no swab mh: swab 2, 5, 10, 15, 30: swab capacity 2 ml, 5 ml, 10 ml, 15 ml, 30 ml。
Rationale
The active preservation fluids embedded in the sample tube contain high-efficiency viral fission fluids, such as salt (sulfate cyanide, sulfate, etc.), capable of rapidly disassembling viral proteins and rendering the virus inactivated, thereby significantly reducing the risk of secondary infections among medical and transport personnel. The addition of rnase inhibitors in the liquid at the same time prevents the degradation of the nucleic acid, ensures that the nucleic acid of the virus is kept and transported at constant temperatures for a certain period of time, and facilitates subsequent pcr testing, especially for large-scale screening scenarios。
The virus sampling tube, also known as the specimen transport tube, is used by the cdc, the clinical department, and the laboratory for the monitoring of raw microorganisms of infectious diseases. For new caps, influenza viruses (common flu, highly pathogenic avian flu, influenza a, h1n1, etc.), avian influenza viruses, hand-foot mouth viruses and other types of viruses. It is also used for sampling of husks, chlamydia, chlamydia, etc。
Virus sampling tubes can also be used to transport tissue samples from sampling sites to testing laboratories that swab or specific locations, pending extraction and testing by the pcr and necessary cell culture。
Materials
Medical pp polypropylene
Composition
Seal cap + tube + non-activated preservation fluid + label bar code, etc。
Specification model
Mvtm-2a (fmh), mvtm-5a (fmh), mvtm-10a (fmh), mvtm-15a (fmh), mvtm-30a (fmh), mvtm-2b (fmh), mvtm-5b (fmh), mvtm-10b (fmh), mvtm-15b (fmh), mvtm-30b (fmh)。
A: swab for delegates b: swab for delegates fmh: swab for non-active 2, 5, 10, 15, 30: representative tube capacity 2ml, 5ml, 10ml, 15ml, 30ml。
Rationale
Non-activated virus preservation fluids do not contain fissure solutions, but solution components that are formed by multiple host cells of the virus, do not damage the structure of the virus, maintain its activity and infectivity, follow-up can be used not only for the detection of the virus's nucleic acid extraction, but also for the development of the virus's separation activity, but for the detection of nucleic acid, care should be taken that the nucleic acid extraction should be purified after it has been eliminated, with strict protection。
Web site: https://www. Medial-swab. Com/, http://cn. Medial-swab. Com/, http://cn. Medicoswab. Com/, http://www. Caiyangshizi. Com/