Chlorine is a good organic solvent that dissolves various organisms (including grease, wax, etc.) and also dissolves high molecular materials such as organic glass, resin, paint, rubber, etc., which can be used as adhesives. Trichloromethane is also an important feedstock for organic synthesis, for the manufacture of cfc-22 and tetrafluoroethylene, as well as for the extraction of penicillin, perfume oil, bioalkalis, etc。
"rurality."
Chlor chloroform is a compound formed by the replacement of three hydrogen atoms of methane by chlorine atoms, also known as trichloromethane, also known as “coloxin”. Chemical chcl3. Liquids that are colourless, transparent, volatile, non-combustible and slightly sweet. Equivalent. Relative molecular mass 119. 38. Relative density 1. 4984(15°c). Melting point - 63. 5°c. Boiling point 62°c. Insoluble in water: 1. 062 at 0 °c, 0. 895 at 10 °c, 0. 822 at 20 °c, 0. 776 at 30 °c, capable of mixing with organic solvents such as ethanol, ether, oil ether, benzene, carbon tetrachloride and carbon dioxide. Photogas (cocl2) that is highly toxic to air oxidation by light. A ethanol stabilizer of 1-2% can be added to chloroform to convert the generated photogas to diethylene carbonate, thereby eliminating toxicity. Anaesthesia, which used to be used as anaesthesia, stimulates mucous membranes and changes faster from a state of excitement to fatigue, dizziness, anaesthesia and can be fatal in serious cases. They are no longer used for anaesthesia because of toxicity, but are still commonly used as solvents in the chemical industry such as pharmaceuticals, plastics, oil additives and organic synthesis. They are common pollutants in the atmosphere and in the water environment. 1. 9 μg/l concentrations increase the risk of cancer by 1 in 100,000. Closics should not be present in drinking water, but chlorinated disinfection of drinking water containing organic pollutants often produces chromatographic concentrations. Therefore, the use of ozone-sterilisation in drinking water is more reasonable for hygiene purposes than chlorination. The maximum acceptable concentration in air was 50 mg/kg。
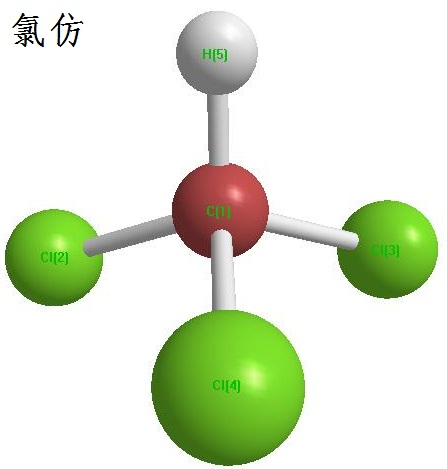
Figure 1 shows the chemical structure of chloroform。
Chemical reaction
Trichloromethane is chemically active due to the induced effects of three chlorine atoms. Easy hydrolysis, production of alphaic acid and hydrogen chloride; in the case of alkaline catalysts, additives can occur, e. G. Chlorine and acetone at 50 °c at potassium hydroxide; in alkaline solutions, trichloromethane reacts with bethamine, which forms thallium, which has a special odour to detect its presence. Further chlorination of trichloromethane can generate carbon tetrachloride。
Trichloromethane reacts with phenol in alkaline solutions to produce hydroxyl aromatic formaldehyde。
Trichloromethane reacts with benzene in the catalytic effect of aqueous aluminium trichloride, resulting in triphenyl (gb) methane. This is an important method of preparing triphenyl methane dyes。
In light conditions, chloroform can be highly toxic to oxygen oxide in air. Sealed in brown bottles. Where partial oxidation occurs, 1% ethanol may be added to destroy photogas (generation of non-toxic diethylene carbonate)。
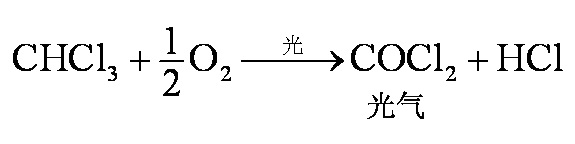
Figure 2 is the photogas from which the chloroform has become highly toxic in light conditions
Use as a telomer in alkyl polymer reaction. The peroxide triggers an argon molecule that can produce hundreds of thousands of polymers and, if added to chloroform, can keep the acoustic molecule number below 10 or more, known as a regulated polymer reaction, so that the polymer reaction can proceed as required. It is also an important organic synthetic reagent. Under the action of alkaline, alkyl reaction produces halogenated three-member ring compound, which is an important method of synthesizing three-member ring compound. Oxidizes into highly toxic photogas under the light, in the form of 2chcl3+o22cocl2+2hcl。
Product quality standards
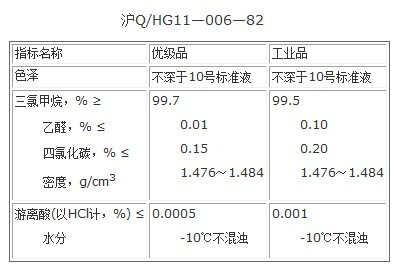
Figure 3
Anesthetic
Chloroform has historically been used as anaesthesia in surgical operations. In 1847, british doctor simpson was looking for better anesthesia, and the french chemist duma sent him a drug that he and his two colleagues had tested in person, volatilizing a small, sweet and special smell of gas, which they then fell asleep. That's chloroform, and it was found that it could be anaesthetic. In return for the successful discovery of dangerous experiments, it is fortunate that the initial trial was conducted in an appropriate amount and that no accident had occurred. Simpson then finds out the amount and efficacy of the drug and applies it to clinical purposes. It has been found to be somewhat toxic to the liver, kidneys and possibly carcinogenic and rarely used。
"the purity of chloroform."
(1) photogas and free chlorine testing
1) photogas testing: photogas is soluble in benzene and toluene, insoluble in cold water, hydrolytic in hot water, and smells particularly bad. 2 ml of chloroform from the test tube with 3 drops of distilled water and 1 drop of silver nitrate solution (0. 2mol/l). The presence of light in the event of white confusion。
2) free chlorine testing: the presence of photogas can be indirectly demonstrated through chlorine tests because of the generation of chlorine gas and carbon monoxide following photogas decomposition. 3 drops of chloroform, 5 drops of distilled water and 1 drop of potassium iodized solution (0. 5mol/l) in the test tube. The presence of free chlorine in chloroform is illustrated by the fact that it becomes rose or purple as a result of iodizing。
(2) distillation purity
Chloroform contains impurities such as water, ethanol, photogas and free chlorine. The photogas is washed first with sodium sodium phenol, ethanol is washed with water and 5 to 6 thorium sulphate, half of the volume of chlorine in each case, and 5% of the volume of chlorine in each case. It is then washed with sodium hydroxide rare solution twice, water 2-3 times, then dehydrated with calcium chloride without water (or sodium hydrosulfate, potassium carbonate), distilled at a rate of 1-2 drops/s, collected from 60 to 62°c and stored in a brown grinding reagent。
If the distillation method is not capable of removing organic impurities, it may be used to extract pure carbon。
The method of preparation
Industrial chloroform can be obtained by methane chloride or by ethanol or ethanol and sodium hypochlorate:
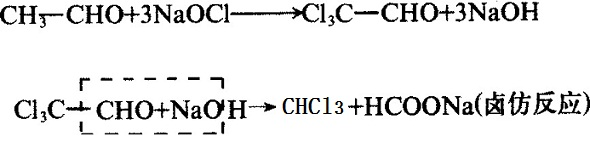
Figure 4 figure
1 based on acetylene, in alkaline solutions, it reacts with calcium hypochlorate and can be produced as chloroform. Acetone can also be used as a feedstock to react to calcium hypochlorate。
2 methane as feedstock, direct chlorination. This response is a strong heat-discharge response to increase the yield and reduce the by-products. Methane perchloromethane, dichloromethane and carbon tetrachloride) should be strictly controlled in molby and chlorine, while at the same time controlling the reaction temperature, even if it could not be a single product, although production rates were higher。
3 hexachloropropanone reacts with sodium hydroxide, available. Cl3coccl3+naoh→cccl3coona
4 carbon tetrachloride as feedstock, which reacts with hydroxide。
5 carbon tetrachloride as feedstock that reacts with ethanol in ultraviolet light. Ccl4+ch3ch2oh→+clch2ch2oh
6 carbon tetrachloride, acting as a catalyst zinc powder, is reduced by hydrogen gas and is comparable to chlorine。
Dangerous situation
(1) vapour is toxic and inhalation causes poisoning. It's anaesthetic. Ingestion or prolonged inhalation causes death. The u. S. Food and drug administration prohibits the use of food, medicine and cosmetic packaging, including cough medicine, toothpaste, etc。
(2) non-flammation, but long-term exposure to flames or high temperatures that can burn, and highly toxic and irritating smoke. Photogas is toxic when exposed to sunlight, oxygen, humidity, especially when exposed to iron. Hydrogen chloride and photogas can be produced at high heat. The acceptable concentration in air is 10 ppm (50 mg/m3) in the united states and 50 ppm (240 mg/m3)*。
[invasive route] chloro chloroform is rapidly absorbed from the lung and is widely distributed throughout the body, either through digestive tracts or undamaged skin. 30-50 per cent of inhalation after cessation of exposure is excreted in prototyre with excretion in 15 minutes, with small parts decomposition in the body and excreted in the form of chlorinated compounds in urine. Vapour inhalation, ingestion, contact with skin and eye。
Neural system, liver, kidney, heart, eye, skin。
[calmic poisoning and harmful effects]
Chlorine is irritated to the skin, with a sense of burning, followed by red spots, oedema, bubbles, and can cause dry skin and cracking without permanent damage。
At high concentrations, chloroform has a strong anaesthesia, stimulates mucous membranes and moves faster from a state of excitement to a state of fatigue, dizziness, anaesthesia or death. Unintended deaths caused by the use of chloroform as an anaesthesia are generally due to damage to the liver and heart. Short-term exposure to chloroform causes dizziness, fatigue, digestive disorders, mental retardation and even coma. Chronic overdose can cause liver and kidney damage. Alcohol users can make the effects of chloroform faster and more severe. Hepatic dysfunction is more pronounced than depression or kidney damage in the central nervous system due to severe exposure. In general, chronic poisoning of chloroform is manifested mainly in vomiting, ingestion, reduced appetite, weakness and mental abnormalities among the severely affected. Chloroform is a known carcinogen。
It produces light gas when it burns, especially when there is a fire。
First aid
Treatment consists mainly of treatment for symptoms. Acute poisoning prohibits adrenaline. Added to storage 1-2% ethanol to produce a non-toxic ethyl carbonate from photogas and ethanol
Co(oc2h5)2
If the chemical enters the eye, it is washed with water immediately; if it is exposed to skin, it is washed with soap and water quickly; if it is inhaled in large quantities, it is immediately removed from the scene to fresh air, in serious cases, by oxygen or artificial respiration; if it is swallowed by error, vomiting, gastric washing, and if it is serious, without vomiting, it is immediately taken to a hospital for treatment。
[protective measures] production equipment shall be closed, the site of production shall be better ventilated and operated with enhanced personal protection, appropriate working clothing and protective glasses. Workers whose skin is wet or contaminated should be washed quickly. Permeable work clothes are removed quickly if they are wet or contaminated。
Pre-employment and periodic medical examinations should include examination of liver and kidney functions, with special attention to the history of the nervous system, skin diseases and alcohol poisoning. The detection of excretion and blood chloroform levels helped to identify acute exposure。
Method
(1) measurement in air: active carbon adsorption, cs2 treatment, gas chromatography analysis。
(2) measurement in water: analysis by gas chromatography or gas chromatography-mass spectrometry。
[store] filled in glass bottles or metal drums with 5% waterless alcohol as a stabilizer. They are stored in cold, dry and well ventilated places. Separated from strong-alkali items. In order to prevent the generation of photogas, light avoidance and insulation should be avoided。
(b) burning after mixing with other flammable fuels is preferable. Care must be taken to ensure complete combustion in order to prevent the generation of light. The installation of acid scrubbers is necessary to remove the hydrogen halogen acid produced. Whenever possible, they should be recycled and sent back to the supplier by distillation。
[use attention]
(1) closics are organic drugs whose main poison is the central nervous system, which has anaesthesia and can cause damage to the human heart, liver and kidney. (2) it should be noted that chloroform can form photogas in ultraviolet light or high heat, which is highly toxic。
(3) the maximum permissible concentration in air is 50 ppm。
(4) the fire may be saved by sand, fog, carbon dioxide。
[reference]
Rhomingquan, ed. . Manual on common toxic and hazardous chemicals. Beijing: china light industry press. 1992. 382-383。
Yuan, guangming, editor-in-chief, the social dictionary of science and technology hangzhou: zhejiang educational publishing house. 1992, p. 223。
Http://www. Chemicalbook. Com/producingchemicalpropertiescb54133133. Htm
Http://baike. Baidu. Com/view/323925. Htm




