From molecular orbital theory to front-line orbital theory to straight-chain co-polymeric molecule orbits, we have invested a great deal of time and energy in exploring the theoretical basis for a perigramic response. Today, we can finally move to a new phase in which we can begin to explore the specific types of response to the responses。
In previous studies, our main focus has been on ion-type reactions in organic chemistry. In these reactions, electrons move from an atom with a negative charge to an atom with a missing electron, usually involving an anion or anion as an intermediate for reaction. For example, the friedel-crafts reaction, which we have studied earlier, and the pro-nucleus reaction of gypsy reagents with compounds such as formaldehyde, ketone, are typical examples of such reactions。
The circular response is a unique type of organic response. Unlike the ion-type reactions that we have studied before, electrons in circumference-reaction flow around a ring, with the break-ups of old and new keys occurring at the same time, without any intermediate generation being involved. Such reactions do not require the involvement of metal catalysts or oxidants, but are accomplished by electronic transfers and re-allocation within molecules. The perimeter response is divided into three main categories: electro-cyclic response, ring-acceleration response and consideration of migration response. Today, we will begin with an in-depth understanding of the mechanisms and characteristics of the electro-cyclic response。
01
Definition of looped response
In light or heat, the electron ring of two carbon at the end of acrylic acrystasy is synthesized into a single consideration, resulting in a double-key acrylic acne less than the original molecule, or its reverse reaction - the acoustic acoustic accelerator - is converted to acrylic acne acne, which is commonly referred to as an electro-cyclic reaction。
02
Concepts related to electro-cyclic response
As we discussed earlier, the core of the organic response is electrostatic attraction and orbital overlap. By definition, we can look at the mechanisms of circumference from the perspective of orbit overlap. In addition, in the study of reaction mechanisms, the front-line orbital theory states that the most critical molecular orbit in the process of electrocyclic reaction of cosmololeene molecules is their highest occupation molecular orbit (homo). Thus, when we look at the electrocyclic response, an effective starting point is the mapping of the homo co-polymeric, which will help us understand the dynamic processes and decisive steps of the response。
For ease of discussion, we will use 1,3-5-hexatrile as an example. The top occupant orbits of 1,3-5-hexadiene have been described in detail in previous discussions and are therefore not repeated here。
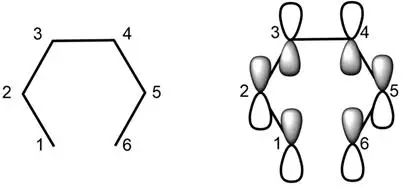
Figure 1
2. 1 circumference and spin
The closing steps in the electro-cyclic response involve the end carbon atoms of co-alkyl, with a new consideration of its electron interactions. If carbon 1 and carbon 6 in figure 1 overlap directly with each other, they form a molybdenum key. However, to form the key, the p-track of carbon 1 and carbon 6 requires a specific flip。
In the electro-cycling response, this reverse process is usually described as “rotation” and “rotation”. The spin refers to the rotation of the keys on two carbon atoms in the same direction, which can be further subdivided into a spin-off and a reverse-clockwise direction. The rotation refers to the reverse direction of the keys on the two carbon atoms, which can be divided into the internal and outward rotations。
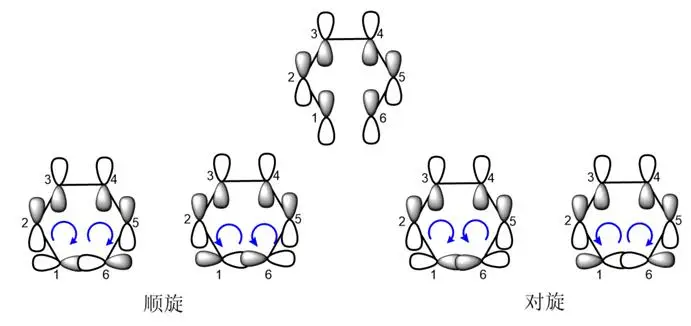
Figure 2
2. 2 “permissive” and “prohibited”
Whether it is a spin-off or a spin-off, there are two different results in the electro-cycling response: one is overlap, which can result in a key track; the other is overlap, which can result in a back-key track. In the discussions surrounding the response, we have specific terms for these overlapping approaches. We will refer to overlap as “symmetric permissibility” and overlap as “symmetric interdiction”。
It is important to note that the terms “permissible” and “prohibited” are thermodynamic concepts that consider the possibility of reaction from a dynamic energy perspective. The so-called “stop” means that the reaction is more dynamic under a synergistic mechanism, making it less likely to occur, but this does not mean that the reaction is not possible under other mechanisms. “permissible” means that the response is less dynamic and therefore more likely to occur under a synergistic mechanism。
03
Cyclical 3d selectivity
Whether it is a spin-off or a spin-off, as long as the conditions for “symmetry permit” are met, the reaction can proceed smoothly to form a ring compound. In cosmopolitans where there is no substitute, only one product is usually obtained as long as the reaction is “symmetrically permitted”。
However, the situation becomes more complex when there is a replacement base for carbon at the end of the carbon atom of co-polypolymer. Different methods of rotation (continuation or converse) lead to different stereochemical results, thus affecting the stereoselectiveity of the product. This means that the presence of a replacement matrix has diversified the choice of response pathways, which may lead to the formation of multiple stereoisomers。
So how do we draw the structure of the product with a stereoselective structure? A simple step can be followed to map the structure of products with stereoselectiveness。
In the case of 1,3,5-hexatriolene with methyl-replacement radicals, we can proceed according to the following steps: 1. Mapping of the front-line orbit homo: highest occupation molecular orbital (homo) of 1,3-5-hexatriolene parent nuclear, with special attention to the p-orbit of the carbon atoms at the end. 2. Marking replacement matrix: methyl replacement matrix in the appropriate position according to the reactor configuration. 3. Determination of end-orbital rotation: on the basis of the rotation of the end-orbital mode (continuation or symmetry), determination of which rotation is symmetrical and, on that basis, the product structure is stereoselective. Through these steps, we can predict and map the products of stereoselective responses。
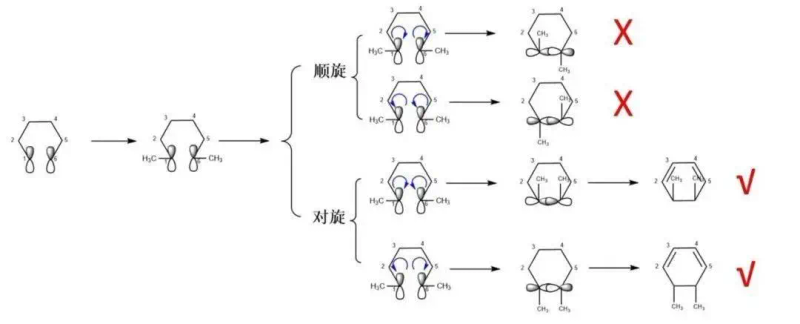
Figure 3
If we seek to acquire the transmutation, the key is to adjust the characteristics of the homo. Specifically, we can only expect the opposite shape when the last wave phase of the homo changes. How, then, can this shift be achieved
03
Light reaction in the electro-ring reaction
If we aim to acquire a reverse product, one effective method is to induce the molecule to leap away from its basic state to its stimulating state by applying appropriate light to it. In this process, the previously basic homo will be converted into a lumo (lowest unoccupied molecular orbit) in a stimulating state. Through this shift, we can influence the electro-delaying and reactional activity of molecules and thus have the possibility of acquiring the required transgenic product。
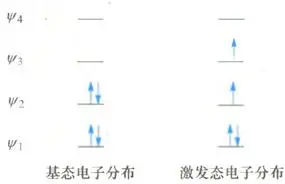
Figure 4
We have already given a detailed account of the specific methodology for the mapping of the luminium molecule orbit in lumo (minimum unoccupied molecular orbit). Then, following the established steps, we can portray the product of the reaction, at which point the result is often a reverse configuration. This phenomenon is the manifestation of the electro-cycling response in light conditions。
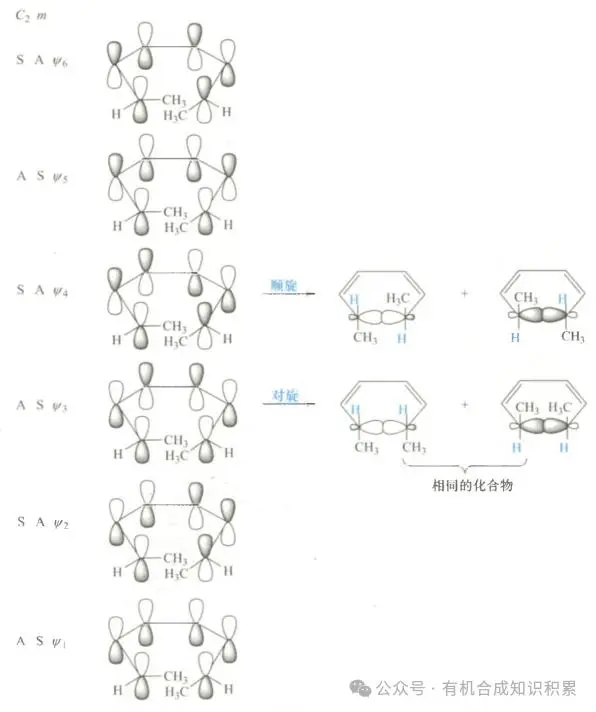
Figure 5
By cleverly using light to induce molecules to leap away from their underlying state to their stimulating state, and thus to change the routing of their lumo molecular orbits, we can direct reactions in the direction of producing a contrario. This process not only demonstrates the practical application of molecular orbital theory in chemical reactions, but also provides us with a powerful tool for understanding and controlling the stereochemicals of organic reactions。




