In the field of polymer science, precision regulation of monosequences and microstructures is key to determining the thermal, mechanical, crystallization and optical properties of materials, yet the development of synthetic methodologies to achieve this goal has always been a major challenge. Traditional grubbs-type resorption catalysts are often difficult to distinguish effectively in the face of high-reactive alkyl, leading to disorderly aggregatement or frequent by-reaction (figure 1a). To address this challenge, scientists usually rely on sophisticated mono-design strategies, such as introducing reactive alkyl pairs, or using inert combinations of officials to achieve alternation (figure 1b). These methods, while somewhat successful, undoubtedly add to the complexity and limitations of synthesis。

In recent days, a new asymmetric, chained open-ring/cross-separation amalgamation methodology has been successfully developed by a group of deputy researchers from the shenzhen glabbs institute of science and technology (figure 1c). The method tactfully uses an anion-type, 14 electronic handmade grubbs catalyst to overcome the challenge of simultaneously achieving a precise intersynthesis of high-contensive cyclopropane with high-reactive end-of-state olefin in the same monomer. The polymeration process is coordinated by two sequenced re-decomposition reactions: first, the dissolvation of the ring-driven ring acrete, and then the cross-decomposition of the terminal alkyl by sequestering control, resulting in the creation of a sequenced, fully chloroolefin structure and a manual centre with highly selective polymers. Paper entitled “sequence- and stereocoNotroled ring-openning/cross mThe topic of "etathesis polymerization" is published in jacs。
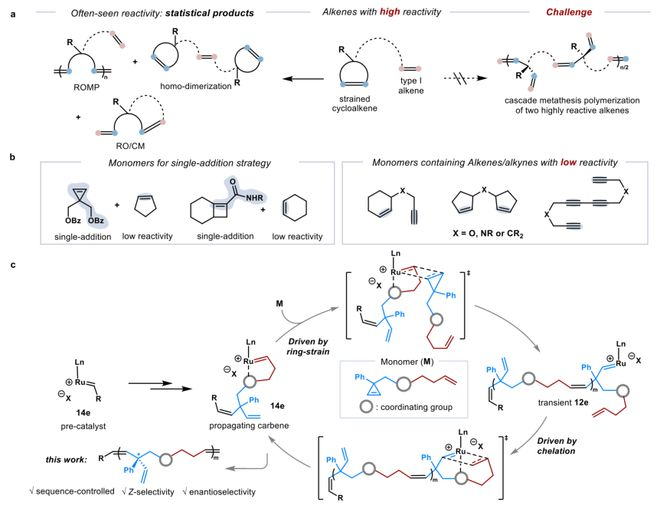
Figure 1. Serial-regulated chain decomposition polymer design with high-reactive algae monomer. A the chemical selectivity between ring-strengthed and highly reactive i-end-of-pipe-olefin in typical recomposition reaction is poor. B mono-design policy based on a combination of (left) and (right) cascaded decomposition. C work: sequence regulation of the catalyze of the anion, z-selective, asymmetrical openings/crossing decomposition aggregation。
In the course of the study, the team first designed and synthesized a cyclopropylene-pentane monomer (m1-m5). Through systematic screening of a range of catalysts (figure 2a), they found that traditional grubbs catalysts (e. G. Grubbs-ii, hoveyda-grubbs) produced by-products only and could not obtain target polymers. The key breakthrough was the use of the anion effect to regulate the electronic nature of the centre. When using an anionic koji-grubbs catalyst ru-3 with a strong-sorting electron trifluorosulfonate root, m1 aggregates exhibit a very high chemical selectivity, with a production rate of 79 per cent for target polymer p1, with an average molecular mass of 35. 7 kda and a dispersion of 1. 64. To further optimize the catalyst, polymer efficiency is maintained when using ru-5 with a greater degree of resistance to nhc formulations, while the hand-centre of duplicate units in polymers has an excess of up to 95 per cent of the map (figure 2b). An mri hydrogen spectrometry confirmed the alternation structure of cyclopropylene with the tailings of the alkyl in the polymer, and no congener 2c was observed。
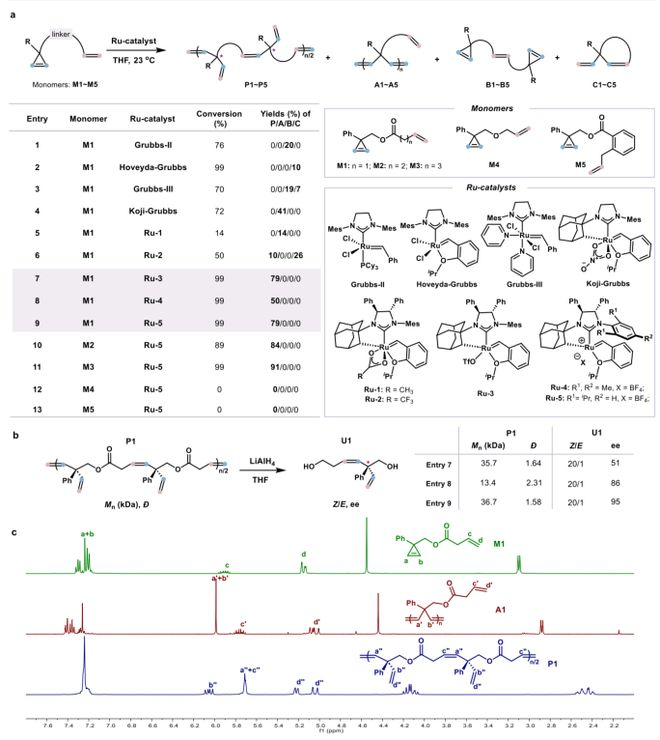
Figure 2. Development of asymmetrical z-selective chain decomposition aggregates containing cyclopropylene and end-of-stage olefin monomer. A screening table for optimal catalyst. General conditions: monobody (0. 1 mmol), monobody/catalyst = 200:1, = 0. 1 m. For entries 1-6, the production rate is measured by the crude mri hydrogen spectrum; for entries 7-11, the production rate is the rate of the separated polymer. B gpc analysis for polymer p1 and z/e-selective and transversal modules. C. Analysis of the hydrogen spectrum of the magnetic resonance resonance of a complex two-unit group in p1。
The tectonic integrity of polymers is closely related to manual optical activity. Using a carbon-13 mri spectrometric analysis of p1 with different map overvalues (figure 3), researchers found that when the hand-centreee value rose from 51 per cent to 95 per cent, the previously divided carbon signal peaks merged into a single sharp peak, indicating that polymers had a high structural integrity. Cubic two-colour spectra further confirms the optical activity of manual polymers: 95% ee (r)-p1 and 93% ee (s)-p1 displays a perfect mirror image of a cd signal, while the signal of its low-ee counterpart is significantly reduced (figure 4). It is noteworthy that, although the value of the centre's ee has little impact on thermal stability of polymers and the glassing transformation temperature, this is due mainly to the distance of the centre's space on the main polymer chain。
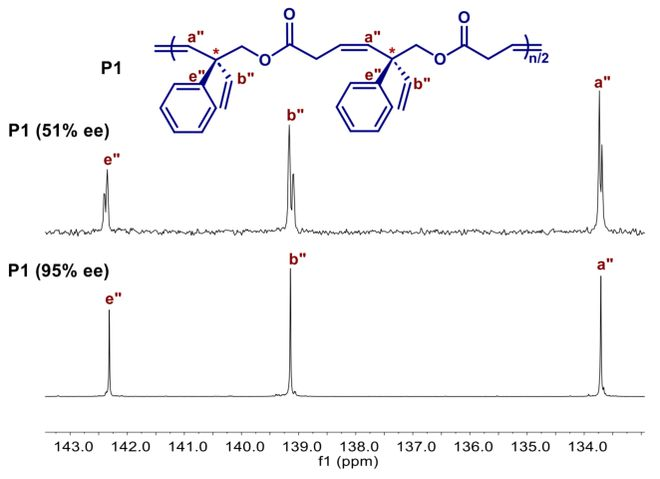
Figure 3. Comparison of the carbon-13 mri for p1 with 51 per cent ee and 95 per cent ee at the handicraft centre。
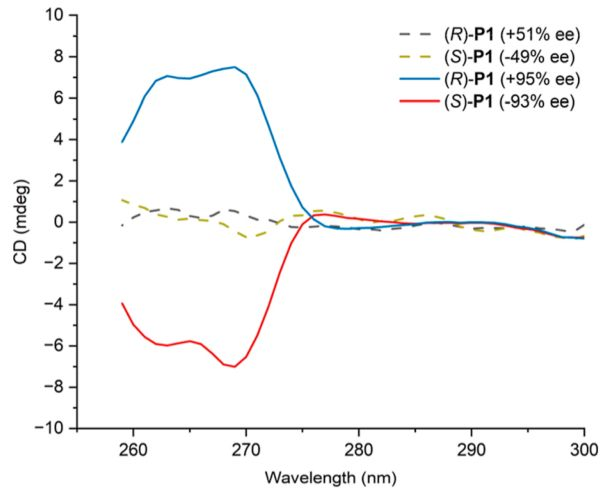
Figure 4. Round two-colour spectral stacking of 51% (r)-p1, 49% (s)-p1, 95% (r)-p1 and 93% (s)-p1 in the handicraft centre。
The team examined the effects of monomer concentrations and catalyst use on polymerization processes for in-depth exploration of polymeric mechanisms (figure 5a). The results show that the molecular volume of polymers continues to grow over extended response times until the single body is depleted and then the molecular volume in parts of the system rises again. Through the maldi-tof mass spectrometry (figure 5b), the duplicate unit of polymers was identified as 214 da and the possibility of circular polymers was excluded. Mark-houwink viscosity test (figure 5c) further confirms the linear structure of the resulting polymer. A study of the mechanisms (figure 6a) shows that the convergence process presents a complex dual mechanism: initial chain growth aggregates, followed by a complete transformation of the monomer, can lead to a gradual convergence between the polymer chain through inter-chain openings/cross-decompositions, resulting in a further increase in molecular mass (figure 6b). Dft calculations also show that this intra-mixed reaction path by sequestering controlled molecules is more energy-friendly。
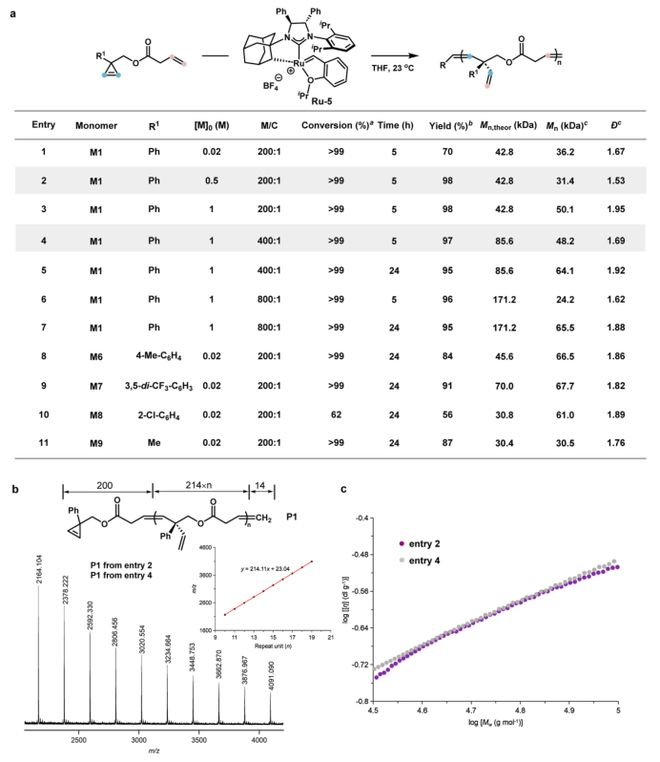
Figure 5. Optimization of serial decomposition aggregations. A the table shows a study of the effects of zero and monomer/catalyst on aggregates. Calculated through mri hydrogen spectrum analysis. By separation. Measurement of average molecular weight and dispersion through gpc analysis. B p1 produced by entry 2 (0 = 0. 5 m, monomer/catalyst = 200 1: 1) and entry 4 (0 = 1 m, monomer/catalyst = 400 1: 1) has the same maldi-tof-ms spectra, as well as a map of the relationship between the mass ratio and the p1. C comparative p1 characterization viscosity by entry 2 (0 = 0. 5 m, monomer/catalyst = 200 1) and entry 4 (0 = 1 m, monomer/catalyst = 400 1), presented as a mark-howink diagram。
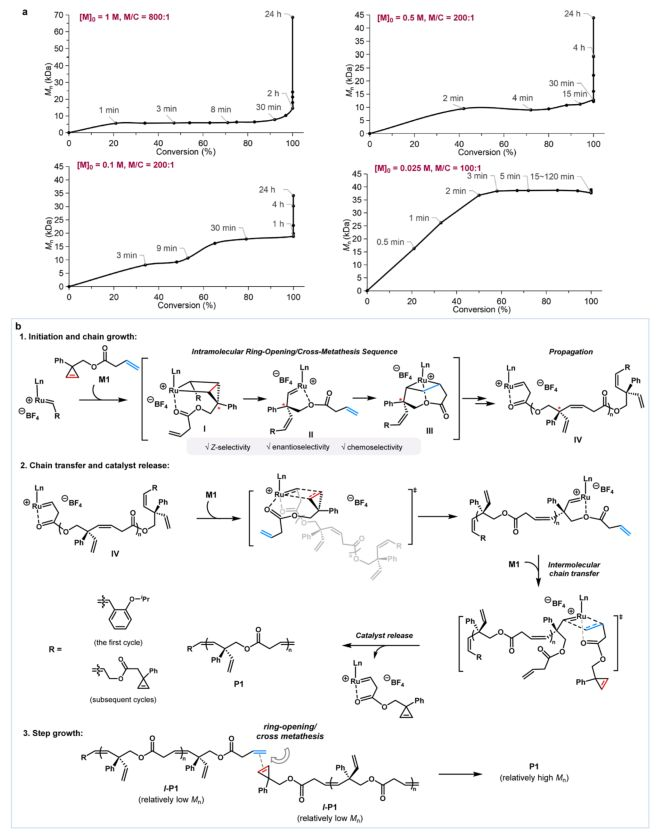
Figure 6. Mechanical study of cascade decomposition aggregation. A map of the relationship between the number of molecules (average number of molecules) and the single-body conversion rate using the r-5 catalyst for m1r. Top left: 0 = 1 m, monomer/catalyst = 800 1; top right: 0 = 0. 5 m, monomer/catalyst = 200 1; bottom left: 0 = 0. 1 m, monomer/catalyst = 200 1; bottom right: 0 = 0. 025 m, monomer/catalyst = 100 1. B the proposed mechanisms for the cascade decomposition of aggregates. For clarity, the structure of ring metalized catalyst ru-5 was simplified。
The study succeeded in achieving precise inter-replacement decomposition of two highly reactive alkylofins through the development of an anion-like grubbs catalyst, which addressed the fundamental difficulty of chemical selectivity in traditional methods. This strategy not only breaks down the limitations of a chain of resynthesis that can rely only on a low-reactive statistical uniform, but also increases to 99 per cent z-selective and 95 per cent symmetrical selectivity, providing an entirely new and universal platform for synthetic sequences and highly controlled functional high-molecular materials that are expected to drive the precise customization and innovation of polymer material performance。




