The principles and applications of the fountain experiment
1. Principles of the fountain experiment
Larger pressure differentials are created within and outside the cylinders in a short period of time, using atmospheric pressure to pressure liquids from the cups under the bottle into the bottle and to form a fountain at the tip of the catheter。
Type of formation of fountains
The following are common gases and absorbents that form fountains。
Gas
Hcl
Nh3
Co2, cl2
So2, h2s
No2
No, o2
(4. 3)
No2, o2
(4:1)
Absorption agent
Water, naoh
Solvent
Naoh solution
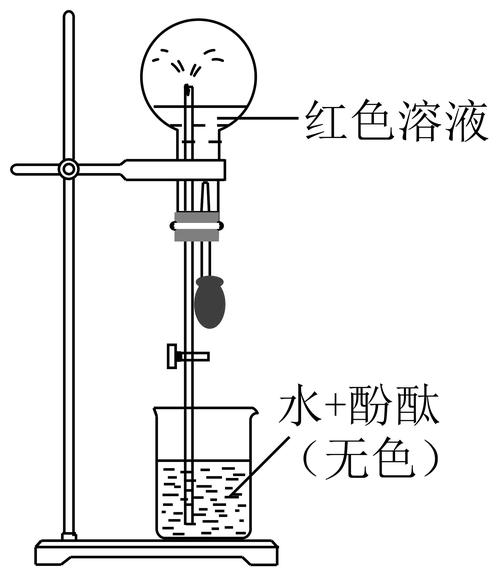
3. Dispersive devices and operating methods of fountain experiments
Fountain experiments are important experiments in high-school chemistry, which by their very nature result in the formation of pressure differentials that cause liquid spraying, with the devices in the teaching materials (dispersion sources) capable of designing a variety of different devices, as shown in the figure, and using different operations (e. G. Solution of gases in water, heat dressing or cooling, generation of gases, etc.) to form fountains。
Please describe the rationale for the design of each device, taking into account its characteristics。
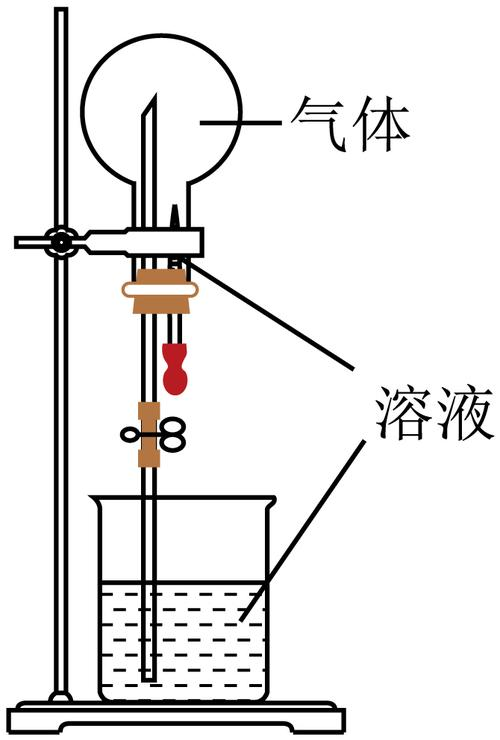
1. The test mechanisms for the fountains are shown in the figure. The following groups of gases - solution - can be used to produce a fountain phenomenon ()
Gas
Solvent
H2s
Rare hydrochloric acid
Hcl
Scarlett
No
Rare h2so4
Co2
Saturation nhocko3 solution
2.
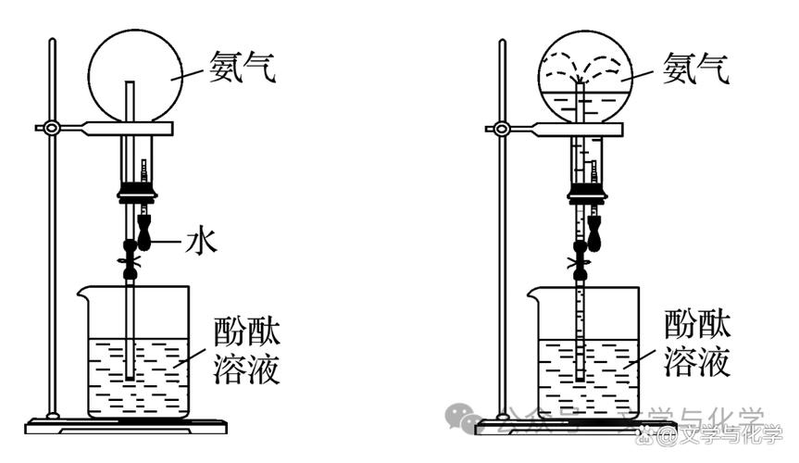
As shown in the figure, the two-colour double fountain experiment is filled with ammonia and hydrogen chloride, respectively, in the flask, with distilled water dripping from the rary solution. The following statement is incorrect: ()
A. In the laboratory, ammonia can be prepared with strong ammonia and lime and hydrogen chloride with strong hydrochloric acid and strong sulphuric acid
B. If c, d, open a, b, there may be white smoke
C. During the experiment, both the water-condensing traps a, c and d can be formed into blue and red fountains, respectively
After the end of the fountain, the solution in the flask is mixed in purple and ammonium chloride solids are available for evaporation
3. A student is actively thinking about other methods of generating fountains and has designed devices as shown in the figure。
(1) in the cone bottles shown in the figure, each of the following substances is added to a sufficient quantity, and the potential for a fountain after reaction is (letter filled)。
A. Cu and rare hydrochloric acid
B. Nhco3 and naoh solution
C. Caco3 and rare sulphate
D. Nh4hco3 and rare hydrochloric acid
The rationale for this method to produce a fountain is 。
(2) placing a sink with alcohol in a chart conical bottle; after water has been added to the tank, the following substances are added in sufficient quantity to produce a fountain. The substance added to the sink may be (lettered)。
A. Enriched sulphuric acid b. Salt
C. Potassium nitrate d. Copper sulfate
The rationale for this method to produce a fountain is 。
The relevant properties of nh3 and hcl can be verified using the devices shown in the figure. Pre-test a, b, c pistons are closed。
(1) if a “spring” phenomenon is to occur in flask ii, a “spring” phenomenon is not to occur in flask i by
。
(2) if a, c pistons are opened and then the glue drip tube is squeezed, the observed phenomenon in the flask is
。
(3) “founding” in flask i by pressing the glue drip tube and controlling the pistons switch, and “founding” in flask ii by
If there's a double fountain in this device, it's done by
。
The principles and applications of the fountain experiment
Think
The device (i) flows a small amount of air into the gas cylinders, pressing a small amount of water into the bottle, resulting in a large amount of ammonia dissolved to form a fountain. Device (ii) saves the glue drip tube and covers it with hands (or hot towels, etc.) thermal flask, the ammonia swells, ejecting the air from the glass catheter, the ammonia in contact with the water, occurs in the fountain (or in the “cooled” flask with ice-impregnated towels, so that the water enters the flask, where the ammonia melts into the water). (iii) adding substances to the sink that increase the temperature of the water, resulting in volatilization of alcohol in a cone bottle as a result of an increase in the temperature and increased pressure of gases in a cone bottle to produce a fountain. The device (iv) introduces a certain amount of h2s and so2 into the flask, which is produced with light yellow powdered matter, with beads attached to the inner wall, and sprays of naoh solution into a fountain. The device (v) turns on one stopper and then closes the stopper when the hcl gas slowly flows into the flask in equal volume, and opens two more until it is fully reacted, observing that the smoke is generated first and then the fountain. In a device (vi), squeeze the glue drip tube and then open two pistons on the upper part of the catheter, smoke is sprayed on the right side of the flask, then open the lower piston of the catheter, and can produce a double fountain。
Pee-ho
1. B
A. H2s and rare hydrochloric acid are not responsive and are less soluble in rare hydrochloric acid and cannot form a fountain; b. Hcl reacts with rare ammonia, and a small amount of thin ammonia squeezed into a round-floor flask can result in a significant reduction in the internal pressure of the round-floor flask and can form a fountain; c. N. O. Does not react with rare sulphuric acid and is very small soluble in rare sulphuric acid and cannot form a fountain; d. Co2 is less soluble in saturated nhco3 solution and cannot form a fountain。
2. D
Nh4cl is unstable, heating evaporation cannot get nh4cl solid, error d。
3.(1) dnh4hco3 and rare hydrochloric acid reactions produce carbon dioxide gases, increasing gas pressure in cone bottles, crushing liquids into flask and forming fountains
(2) a sulphate solubles in water, volatilizes alcohol in a cone bottle, increases gas pressure in a cone bottle and sets alcohol into a flask into a fountain
Parsing (1) cu and rare hydrochloric acid, nhco3 and naoh solution react to release heat, but do not produce gas and do not create greater pressure




