Extra-nuclear electro-leaving and motion i, energy layer, energy level and atomic orbit 1
Capable layers are the layers of extranuclear electrons by energy differences, corresponding to the first and second layers of the atom structure, with symbols k, l, m, n etc. (n=1,2,3,4...). The closer to the nuclear, the lower the energy。
2. Capability level
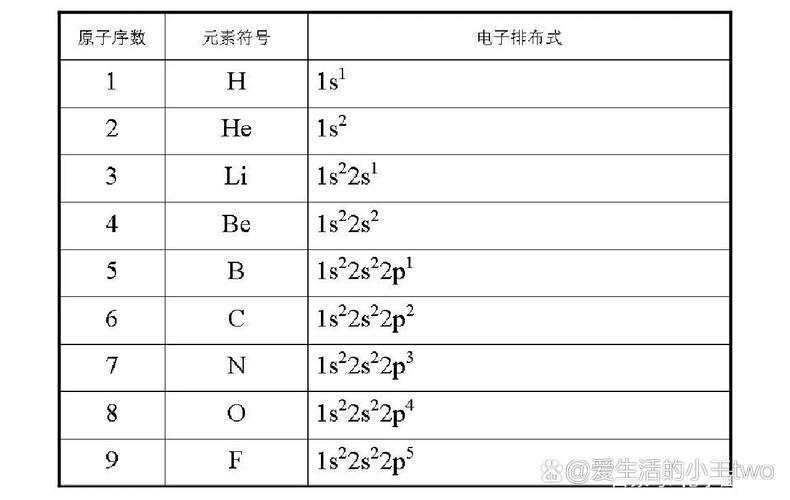
The electron energy of the same energy layer varies slightly and is further divided into different levels of energy, with symbols s, p, d, f, etc. The number of atomic orbits and the maximum number of electrons to be accommodated at each energy level are as follows:
2. The shape, number and energy of the atomic orbit 1. Orbital shape 2. Energy relations iii. Atomic electron release patterns 1. General pattern 2. Three principles 3
Full-filled (p6, d10, f14), semi-filled (p3, d5, f7), full-empty (p0, d0, f0) state is lower and more stable. For example:
Electronic motion and space motion 1. Electronic motion
The movement status of each extranuclear electron varies, with the number of electron = extranuclear electrons. For example, the oxygen atoms (eight electrons) have eight types of electronic motion。
2. State of space movement

Is the number of orbits occupied electronically, the number of space motion = the number of orbits. For example, 1s22s22p4 for oxygen atoms and 1(s)+1(2s)+3(2p)=5 for orbits, thus the number of space motion is 5。
Example 1. Phosphorus (p, no. 15), chromium (cr, no. 24), 3. Iron ion (fe2+, element 26, fe lost 2 electrons) 4. Copper (cu, no. 29), error-proneness and attention order: in electronic layout, (n-1) d can be written in front of ns (e. G. Fe: 3d64s2, not 4s23d6). Loss of electronic order: when transition elements fail electronically, priority is given to loss of ns electronics (e. G. Fe2+ loses 4s2 electrons, forming 3d6). Base and motivating state: the lowest amount of nuclear power in the base state, and the electronic leap to the high energy level to form the trigger state (e. G., the basic li:1s22s1, which could be 1s12s2). The hunt rule exception: full, semi-filled, full-empty and stable, bearing in mind the layout of special case elements such as cr and cu。




