
Sodium carbonate is a "black face" (white tiny crystal), and it's a "crystal."
It's easy to be angry. It reacts with acid, it produces a lot of gas, and it saves the acid! This is better than sodium carbonate! The use is fire extinguisher: foam extinguisher
It likes "cool" and is afraid of "heated" (heat when dissolved, lower temperature, and reaction with rare hydrochloric acid, decomposition of solids, used as dry powder extinguisher, etc.)
Note: sodium carbonate solid is soluble in heat and sodium carbonate solid is soluble in water。
It's softer
It's also a little weird
I. Balance in sodium carbonate solution
Looking at or calculating the five balanced constants, it was found that the balance of the constants was much greater than the other equilibrium constants, and its special nature was highly relevant to the balance. Particular attention should be paid to the ionizing balance of the hco3 - 5。
Ii. Sodium bicarbonate solution ph largely unchanged
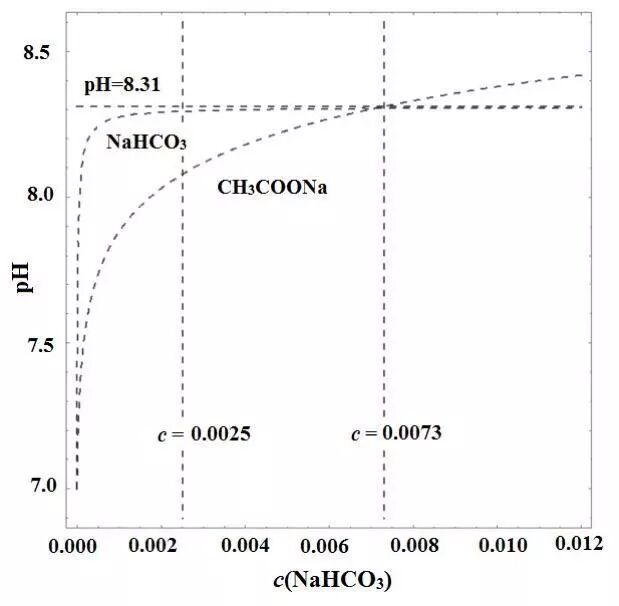
Compared to the weak alkalinity of the ch3coona solution and the nhco3 solution at temperature-to-concentration. The majority of teachers concluded that the nohco3 solution at the same concentration was more alkaline based on hydrolysis. However, as can be seen from the figure, the ph of its solution remained almost unchanged when c (nhaco3) > 0. 0025mol l-1, and when c>0. 0073mol l-1, ch3coona was more alkaline than the nhaco3 solution. This is mainly associated with acidic salts, and it is recommended to avoid comparing the ph size of a weak acid alkali salt to a diaphragm acid alkali salt solution。
Iii. The comparison of the sizes of the particles of sodium carbonate solution shows that, regardless of the concentration of nh-)>c(h+) and c(na+)>c (hco3-)>c (h2co3)>c (co32-) concentrations of other particles vary significantly. In view of the complexity of ion behaviour in the diaphragm acid saline solution, it is recommended to avoid the issue of comparing the various ion concentrations in solutions such as nhco3 and nhso3。
Iv. Cacl2 solution identifies na2co3 solution and nhco3 solution
In secondary school chemistry teaching, the cacl2 solution and the nshamco3 solution are generally considered chemically unreactive, while the cacl2 solution and the na2co3 solution can react and generate white sediment, so that the cacl2 solution can be used to identify the na2co3 solution and the nshamco3 solution. However, there were also chemical teachers who demonstrated that the nhco3 solution was also capable of producing caco3 white deposition with cacl2 solution, which was not considered feasible。
Experimental results and theoretical calculations indicate that the identification of na2co3 and nshaco3 solutions with cacl2 solutions is entirely feasible as long as the soluble content of the samples to be measured is within the normal range of identification (>1%)。
The chemical reaction can be assumed to occur when cacl2 solution is mixed with nhco3 solution as follows: cacl2+2nhh2o+2↑2nacl. This equation has to do with hco3-self-ip。
When both cacl2 and nhco3 have concentrations of less than 0. 1 mol l-1, it may be due to the low volume of co2 produced, which does not allow for the observation of bubbles。
V. Nhco3 solution decomposition
It is well known that sodium bicarbonate solids are thermally decompositionable and that the nshoco3 solution can be introduced in experimental and theoretical calculations to decomposition to sodium carbonate, water, carbon dioxide at constant temperatures or heating. Nhco3 decomposition reacts in solution:
2hco3-==co32-+h2o+co2↑。
Instead of promoting hydrolysis into naoh and h2co3, h2co3 is decomposed into co2 because co2 cannot escape from naoh solution。
Vi. Responses to the participation of hydrate
1 h2o+nalo2+nhhco3=al(oh)3↓+na2co3
2 alcl3+3nhhco3=3co2↑+al(oh)3↓3nacl
3, feso4+2nshaco3 = feco3↓co2↑na2so4+h2o
Convertible to ammonium carbonate: feso4+2nh4hco3=feco3 ↓co2↑(nh4)2so4+h2o
4, 2 mg+2nhaco3+2h2o = mg(oh)2 mgco3↓ + na2co3+2h2↑
Meg reacts rapidly in the nhco3 solution, with h2 and white insoluble。
5. Ca (oh) 2+2nsingco3 = caco3↓na2co3+2h2o
Small amount of sodium bicarbonate: ca(oh)2+nshoco3=caco3↓naoh+h2o
Vii. Reactions from sodium carbonate
1 nacl (saturation) + nh3 + h2o + co2 = nh4cl + nhhhco3
Naclo+co2+h2o =nhaco3+hclo
3, c6h5ona+co2+h2o→c6h5oh (mixed)+nhhco3
4, na2co3 (saturation) + h2o + co2 = 2 nhco3
5. Add rare hydrochloric acid to saturated sodium carbonate solution, and add a bit of mixing, with no gas at first, and the solution becomes sour:
Na2co3+hcl=nshaco3↓nacl; continue to drip the rare hydrochloric acid gas and clarify the solution。




