Sodium napalphate (nhco3), commonly known as small soda, is widely present in everyday life and industrial production as an important inorganic compound. Its unique nature makes it a key player in many areas, such as food, medicine and chemical work, and is the subject of chemical learning and research that cannot be ignored。
I. Basic nature
1. Physical properties
Appearance: white tiny crystals, powdered under normal conditions。
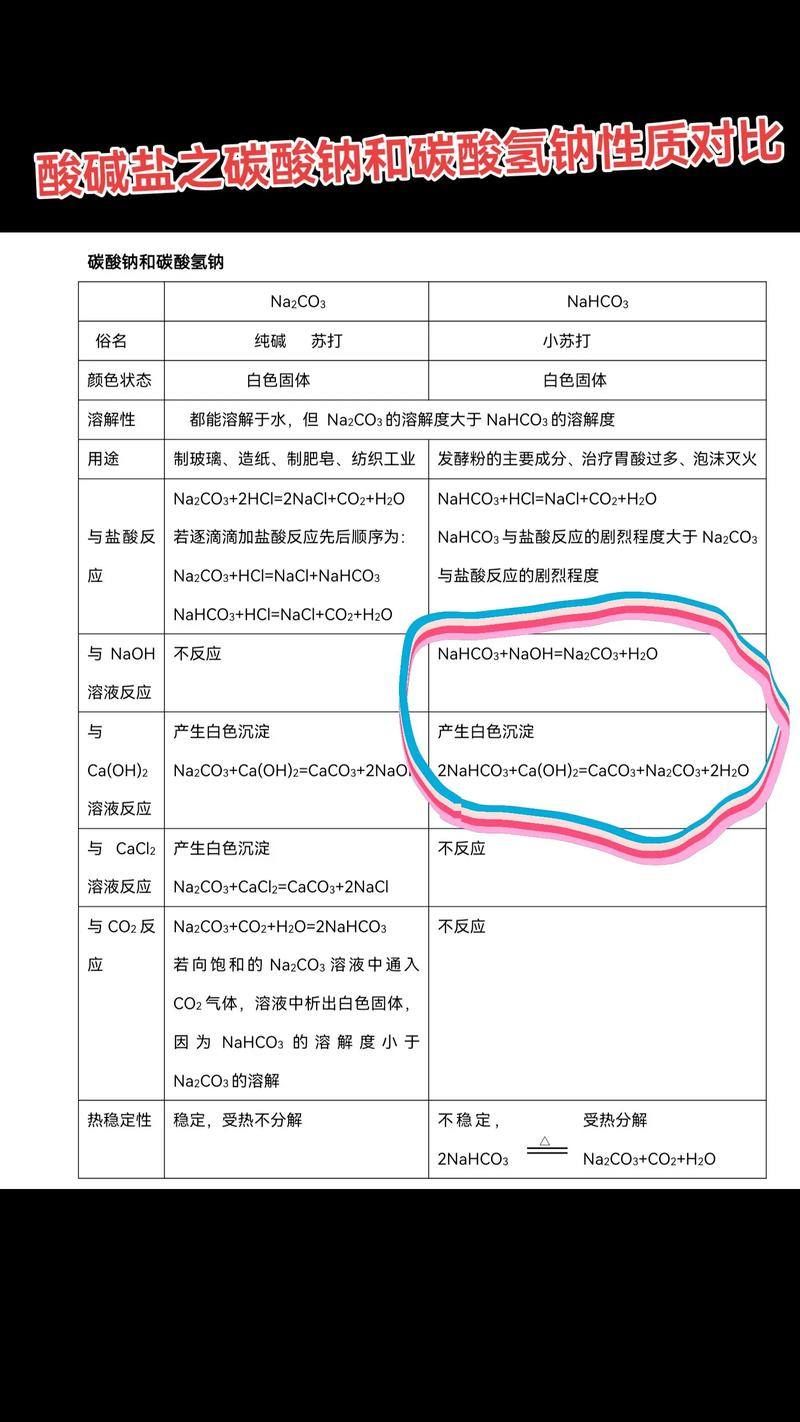
Solubility: aqueous solution is weak in water, with a constant temperature solubility of about 9. 6 g/100g water, which increases slightly but changes little as temperature rises。
Smell: no smell, no smell or a little alkaline。
2 chemical
Thermal stability: sodium carbonate is thermally decompositioned to produce sodium carbonate, water and carbon dioxide, a feature often used in food baking and as an inflation agent to expand and soften the flour。
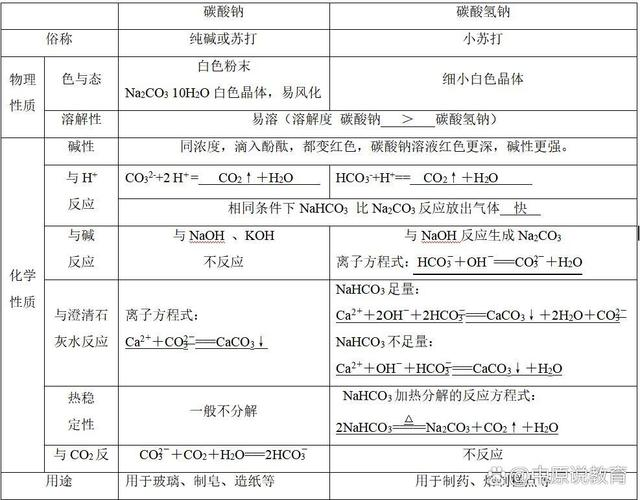
(c) acid reaction: can react quickly with strong acids such as hydrochloric acid to produce sodium chloride, water and carbon dioxide, which is applied in fire extinguisher principles and in stomach acids and scenarios。
Hydrolysis: in aqueous solutions, hydrocarbonate root ion is capable of hydrolysing the solution to make the solution alkaline and ionizing to produce the hydrogen ion, but the hydrolysis is greater than ionizing and the overall solution is weak alkaline。
Preparation methods
1. Industrial preparations

Solway (amino-alkali): using sodium chloride, ammonia and carbon dioxide as feedstock, ammonia is first fed into saturated salt water, then into carbon dioxide, and reacts to produce sodium carbonate and ammonium chloride, and then obtains sodium carbonate products through filtering, washing and drying. This approach, which is the main way for industrial production of sodium bicarbonate on a large scale, has the advantage of cheap raw materials and process maturity, but produces large amounts of calcium chloride sludge with some pressure on the environment。
Alpine method: our scientist, mr. Holdersung, invents synthetic ammonia and pure alkali plants, using ammonia, carbon dioxide (co2) and sodium chloride as raw materials, to generate ammonium carbonate from ammonia and water and carbon dioxide reactions, and to sodium chloride solutions from sodium carbonate and ammonium chloride. The act had improved the utilization of raw materials and reduced environmental pollution, while producing both pure alkali and ammonium chloride products, which were cost-effective。
2. Laboratory preparation
A small amount of sodium carbonate can be made available for sodium carbonate and carbon dioxide and water reactions, and excess carbon dioxide can be fed into saturated sodium carbonate solutions. The reaction results in sodium carbonate, which, because of its relatively small sodium carbonate sodium sodium sodium sodium, is crystallized from the sodium carbonate, which is filtered and dry。




