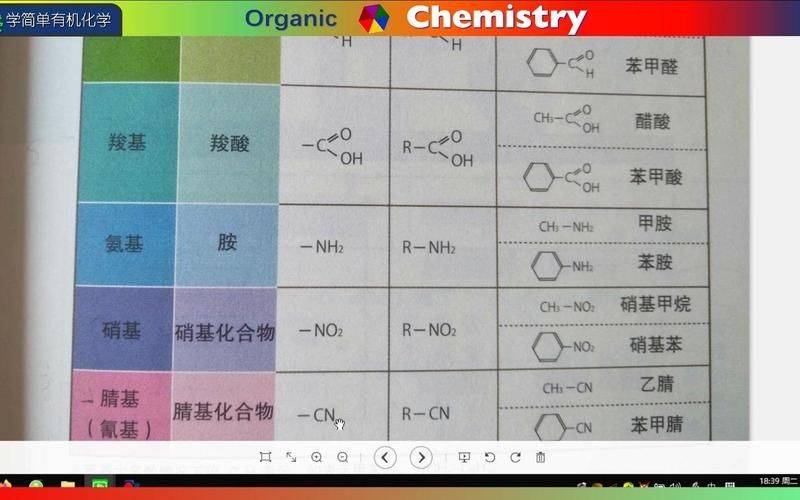
**** *** *** *** / the organic chemical protection corps (ocgc) is a chemical umbrella for the protection of specific service groups in organic molecules to prevent reactions in chemical reactions. In organic synthesis, kpc is widely used to control the selectivity of reactions, prevent the occurrence of secondary reactions and ensure the formation of end-products. In its multi-step response, kpc outlined the protection of specific civilian capacities, which could temporarily block and prevent unnecessary reactions to ensure the smooth operation of the synthetic route. The introduction of kpc and the introduction and removal of kpc require the use of specific reagents and response conditions to ensure that other official groups are not affected. Selective protection groups that increase response play a crucial role in the synthesis of complex organic molecules, and their introduction and removal enhance response selectivity and ensure the synthesis of target products. The kpc classification is classified by reactive activity according to the kpc sensitivity to acid, alkali, oxidizer or reduction agent. Classification by structure is based on the chemical structure of the kpc, e. G., ethers, esters, amides, etc. Categorized by protected official capacity groups, such as the hydroxyl umbrella, the amino umbrella, etc. The protective purpose of hydroxylene is to prevent its involvement in follow-up responses and to avoid secondary reactions. The protective rationale replaces hydroxyl radicals with conservation cells, temporarily rendering them inactive. The kpc features are easy to introduce and remove and do not affect other official groups. The selection of kpc is based on the response conditions and target elements. The common protection group of hydroxyl methylether is the commonly used protection group, which is highly stable and easily introduced and removed. Alkyl ether is also a commonly used protection umbrella, which is stable and can be removed under acidic conditions. Tbt, commonly known as tbdms, has good stability and selectivity and can be removed under alkaline conditions. Acetyl acetyl is a common protection umbrella that is stable and easily introduced and removed. Option 11. Responsive conditions need to take into account the stability of the corps, e. G., whether decomposition or deprotection occurs at high acid, alkali or temperature. 22. The nature of the target molecule, for example, requires the choice of a protection umbrella that is compatible with other sensitive groups of officials if they are present. 33. Conditions of de-protection require the selection of groups that are easily de-protected and do not damage the target elements. 44. The introduction of kpc requires the selection of a mission that can be easily introduced, and the introduction process will not result in a secondary reaction to the target molecule. The introduction of ghp 1 and the selection of suitable protection missions taking into account response conditions and target molecular structure 2 to ensure the efficient and highly selective introduction of ghp 3 to target molecules to protect against unnecessary reactions to ghp in subsequent reactions is an important step in synthetic chemistry that can protect ghb from other reaction reagent attacks. The selection of suitable reagents for removal 1 to control response conditions such as temperature, solvent, response time, etc. Of suitable reagents, according to the type of protection foundation, ensures the complete removal of the protective efficiency 3 monitoring response process and avoids the sub-reaction 4 purified chemical products from the target product, by means of column thawing or re-clining, is a common step in synthetic chemistry, and the selection of suitable reagents and reaction conditions is essential. The removal process requires strict controls to ensure that kpc is completely disengaged while avoiding secondary reactions. After removal, the target product needs to be purified to obtain high purity compounds. Ammonium for the protection of 1 amino-protected ammonia is an important official body that plays an important role in organic synthesis, but sometimes it needs to be protected from other reactions. The choice of the kpc will depend on the specific conditions of the response and the target molecular structure. 3 the introduction of the protection corps is usually achieved through aerochemical or alkylization reactions. The removal of the kpc will require the removal of the kpc after the completion of the synthesis response to restore the activity of the ammonia. The common amphibians protection group, the amphibians protection group, is one of the most commonly used, such as acetamide (ac), pz and tbt. The amphibians are generally removed under acidic conditions, e. G. By trifluoroacetic acid (tfa). Alkyl protection groups, such as the tertbutyl (tbu) and thyl (bn) protection groups, are stable under acidic conditions but can easily be removed under alkaline conditions. Alkyl protection cells are usually used to protect ammonia from reactions under alkaline conditions. The choice of response conditions for the ammonia protection base selects the protection base that is suitable for the particular response conditions to ensure stability during the synthesis process. For example, under strong acid conditions, protection cells for acid stabilization should be chosen. The follow-up response takes into account the need for a follow-up response and selects a protection mission that can easily be removed, avoiding disruption to the follow-up response. The nature of the ammonia is based on the type and responsiveness of the ammonia, and the appropriate protection group is selected, e. G. For belamine, the boc or fmoc protection group. The introduction of ammonia protection groups is a common strategy in synthetic chemistry to protect ammonia from other reactions. For example, the removal of 1-acid conditions from the 1ylization reaction to aminoated 2-alkyl alkyl reaction to amino alkyl alkyl reaction with halogenated alkyl or ethanol uses acids and amino-formation of the 1-acid conditions of the alkyl protection group, e. G. The treatment of boc to protect alkyl 2-alkaline conditions with alkalis to protect, e. G. The treatment of cbz to protect 4-ray conditions with catalytic hydrogenification to protect fmoc, e. G., the conditions for photolysis to remove the nitro-methamphetyl protection to remove the amino-protective foundation depend on the protection-type used and the selection of appropriate conditions to avoid secondary reactions from target molecules. Neptunium protection and protection of the alkyl base are pro-electric and need to be protected from unnecessary reactions in organic synthesis. The introduction of esters, aamides and halogens, which are common in kpc, includes esters. The selection of the appropriate protection mission would require consideration of the conditions of response, the stability of the protection mission and the conditions of release. The de-protection response needs to be carried out in moderate conditions to avoid affecting other official groups. The common protection base 11, esters of thallium is widely used in organic synthesis and is highly stable and easily detached. 22. The aramid protection group is more stable in terms of both acid and alkaline, and is usually used to respond to the need for stronger protective conditions. 33. Halogen methyl esters (halogenated methyl esters) are commonly used for reactions requiring stronger reduction conditions, such as the reduction of metal hydrogenide. Reaction conditions, such as acid alkalineity, temperature, reagents, etc., need to be taken into account when selecting the response conditions for the base group to ensure that the mission is not disengaged during the response. The selection of suitable protection elements by the target elements will need to take into account the structure and nature of the target molecules and ensure that they do not affect other reactions by the target molecules. The selection of a suitable protection umbrella without conditions of protection needs to be considered to ensure that it can easily be removed without affecting the target elements. The introduction of ayl fluoride reaction in the thallium protection group uses ayl chloride or acetic anhydride to produce esters or amides and to protect the thallium. 2-ester reaction esters esters with ethanol reaction to protect esters. Common alcohols include methanol, ethanol, ethanol etc. 3 the formation of ammonium acetate and the reaction of the acetamine form ammonium to protect the aluminum. Common meds include dimethylamine, diethylamine, etc. The removal of the antimony protection corps is a key step in synthetic chemistry. The choice of suitable conditions for removal from protection is essential to ensure the stability and purity of the product. The selection of suitable reagents for appropriate de-protective reagent 2 control response conditions, temperature, time and solvents, based on the type of protection base, may affect the effectiveness of de-protection and the integrity of product 3 monitoring response by monitoring the response process using techniques such as thin chromatography or nmri to ensure complete de-purification of the 4 product and the removal of residual reagents and by-products by appropriate purification methods and the acquisition of high purity target product formaldehyde the reason for the protective protection of geno-ketone is the high likelihood of oxidizing, reducing, adding, etc., and protection is needed to prevent these reactions from occurring so that other synthetic steps can be taken. Common protection strategies protect formaldehyde/ketone by forming a formaldehyde/dehyde, cyclodehyde/dehyde or alkyl ether, which can effectively deter unnecessary reactions. Kpc's choice is to select the appropriate kpc according to specific response conditions and target products, ensuring that kpc can be effectively removed from the follow-up steps. The common protection group dethylamyl for formaldehyde/ketone is applicable to the protection of formaldehyde and ketone and can be removed by acid or alkaline hydrolysis. Formaldehyde protection groups are widely used for the protection of formaldehyde and ketone, which are relatively stable and can be removed under acid conditions. Decone protection groups are used to protect the substance, remove it from acid conditions and stabilize it. The cyclodoxone, which protects formaldehyde and ketone, has good stability and selectivity. The choice of the official nature of the solid selects the appropriate protective mission, taking into account the nature of the other mission in the target molecule。for example, if the molecule contains acidic performance groups, the acid-sensitive protection cluster needs to be selected. The protection umbrella chosen for response conditions needs to be able to be stable under specific response conditions and able to be removed under appropriate conditions. The introduction of the 1-plus reaction of the formaldehyde/ketone protection group with the use of 2 decoholization and ols, such as the gyldoxin, the vittig agent, and the 3-decodone 3-decoholide and the aridyl chloride reaction with the production of the 4-cyclic and diol reaction to the cyclodehyde, is based on a number of methods for introducing the protection group, depending on the structure and response conditions of the formaldehyde/ketone. The selection of the appropriate introduction methods would increase response efficiency and reduce the occurrence of secondary reactions. Decontamination of 1 acid hydrolysis using acidic conditions, such as acid hydrolysed acetylene or decorate, for the plasma plasmodium. 2-bed hydrolysis uses the alkaline hydrolytic protection matrix, such as alkaline hydrolytic platinum or decolytic ketone. 3 reverted de-protection using re-agents, such as lithium hydrogenated, and de-protected umbrellas. The importance of phosphate for the protection of phosphate is an important body of operational energy in biological molecules such as dna and the phosphate skeleton in rna. In organic synthesis, the protection of phosphate prevents degradation or reaction under reaction conditions. Common phps include thallium (bn), tertbutyldimethyl silica (tbdms) and methylenedioxymethyl (mom). The common protective group of phosphate (bnpo3) nitrous phosphate (bnpo3) is a commonly used phosphate protection group that effectively protects phosphate hydroxylene and can easily be removed. Methoxymethyl phosphate (momoto3) is a medium-intensity protective base, usually used to protect phosphate that needs to be removed under moderate conditions. Tetrabutyldimethyl silicaphosphate (tbdmspo3) is a strong protection umbrella that effectively protects hydroxyl phosphate for reactions that need to be removed under strong acid conditions. The choice of response conditions for the phosphate conservation corps needs to take account of the response conditions, such as temperature, ph and solvent, to ensure that the corps is stable under the reaction conditions and is not accidentally removed. The follow-up response needs to take into account the conditions of the follow-up response and to select a protection mission that is compatible with the follow-up response and avoid interfering with its conduct. Select the appropriate conditions for removal to ensure that kpc can easily be removed and that it does not cause damage to the target molecule. The molecular structure takes into account the structure of the target molecule and selects the appropriate protection umbrella to ensure that it can effectively protect the active umbrella of the target molecule. The introduction of active 1 of the phosphate conservation corps uses suitable reagents, such as dcc or edc. 2 the reaction will react to the activated phosphate with the protected substrate and form the phosphate key. 3 purification of the chemical product by means of column dialysis or re-crystalization. The introduction of phosphate protection groups usually requires active phosphate, which then reacts with the target molecule. Common methods of activation include the use of reagents such as dcc or edc. The reaction requires purification to obtain pure products. The removal of 1 acid hydrolysis from the phosphate protection group uses strong acids, such as trifluoroacetic acid (tfa) or hydrochloric acid, for hydrolysis in solvents such as water or methanol. This method applies to most phosphate protection groups, but may lead to secondary reactions. 2-alkaline hydrolysis uses strong alkalis such as sodium hydroxide or potassium hydroxide for hydrolysis in water or alcohol. The method is effective for certain types of phosphate protection cells, but may lead to secondary reactions. 3 ecd uses enzymes such as phosphate diesterase or monoesterase of phosphate to achieve specific deprotective responses. The methodology is moderate, efficient and well selective. Kpc's summary 1 protection corps importance protection corps is critical in organic synthesis to protect the response officer corps from unintended reactions in the course of the response and to ensure synthesis efficiency and purity of the target molecule. The selection of the appropriate protection mission depends on the conditions of response, the structure of the target molecules and the steps of response, and there is a need to consider the efficiency of the introduction and removal of the protection mission and the impact on the response system. 3 the removal of kpc from kpc must select the appropriate conditions to avoid having an impact on other official groups and to ensure the integrity and purity of the target molecule. The ongoing exploration and development of new protection missions, as well as efficient methods of introduction and removal, are essential for improving the efficiency of organic synthesis and simplifying synthetic steps. The choice of a suitable protection mission would require consideration of compatibility with other missions and avoidance of interference. The introduction and removal of response conditions requires the selection of appropriate response conditions to avoid impact on other official missions. Stable choice of a highly stable protection base to ensure that it does not degrade or convert in subsequent reactions. Dismissed from the protection of the mission's efficiency and conditions, facilitating the later recovery of target elements. **** *** *** *** *** *** *** *** *** ***




