The libs electrodes consist mainly of electrochemically active electrodes, conductor additives, glues, fusions, etc., of which the glue is an important part of the libs electrodes, which bind the active substances and conductants closely to the fusion, form the complete electrodes, prevent the discharge of active substances from falling off, stripping off, and enable the dispersing of both active substances and conductors, thus creating a good electronic and ion transmission network and achieving efficient transmission of electrons and lithium ions。
Substances currently used as electrode adhesives include polyfluoroethylene (pvdf), beryllium cellulose (cmc), butzene rubber (sbr), polyethylene ethylene ketone (pvp), polymethylacrylate (pmma), polyacrylate (pan), polypropylene acid (paa), polyethylene alcohol (pva), sodium algae acid (alg), beta-ring polymer (beta-cdp), polypropylene emulsion (la132), polytetrafluoroethylene (ptfe) and functionally-chemical derivatives or monocorporated polymers。
In libs electrodes, the desired glue performance shall include:
(1) chemical and electrochemical stability in a given electrolytic/electrically decomposition system, resistant to electrolytic corrosion, and no oxidation reaction at working voltage
(ii) good solubility, fast and high solubility in solvents, and the required solvent is safe, environmentally friendly and non-toxic, with the best water solvents
(3) a moderate degree of viscosity, which allows for slurry and the maintenance of slurry stability, with strong binding force, preparation of high electrode detached strength, good mechanical performance and low adhesive use
(4) a high degree of flexability, which is resistant to changes in the volume of activated matter particles during the convulsion and libs recharge cycles during electrodes
(5) be able to form an ideal conductive network with a conductor agent, and have good electrodes and conductive lithium ions
(6) sources are wide-ranging and inexpensive。
The present paper summarizes the results of studies in recent years on the adhesive mechanism of the adhesion of the adhesive in the electrode and on the oil and water system adhesives currently commonly used in the electrodes of the libs。
Sticky mechanisms in lithium ion cells
The libs electrode production process typically consists of four steps: mixing materials (including electrode active materials) into solvents to make battery slurry, coating of the slurry on a fusion, drying and voltage. It is generally considered that the libs electrodes contain three components: activated matter particles (am) as an ion and electronic source, electrolytic porespace filled with electrolytes for ion transfer and carbon-adhesive fields (carbon-binder domain, cbd)。
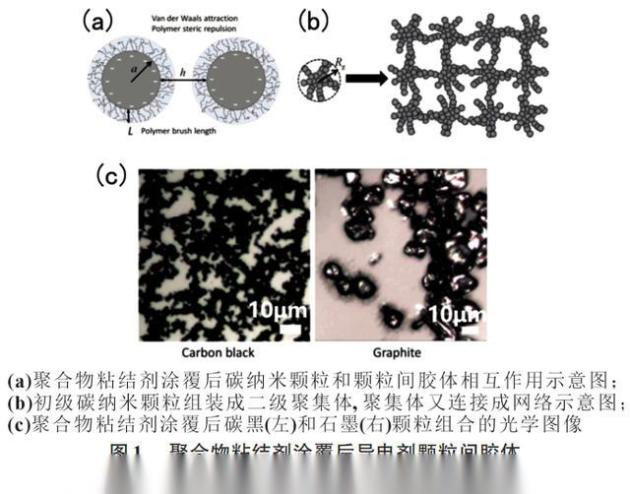
Cbds are usually made of carbon nanoparticles connected to polymer glues (figure 1), and the pre-pulsive plasma required to prepare electrodes consists of micro-metre-grade am particles suspended in cbd, which directly affect the transmission efficiency of ion and electronics in the electrolytes and the quality of passivated layers (e. G. Sei, cei membranes) formed on the surface of active substances in contact with electrolytics, so cbds play a vital role in the process of electrode manufacturing: too few cbds can lead to inadequate electrical connectivity, resulting in poor electronic transmission and insufficient electro-mechanical strength, while too many cbds increase the battery's self-heaviness and volume and even slow down the ion's transmission。
Zielke and others compared the effects of the two cbd models on the surface area, bending and conductivity of the solid electrolyte interface (sei) membrane in the libs charged electrode by means of a new method combining x-ray fault scanning and virtual design. The results show that the content of the cbd has a significant impact on transmission parameters in both the libs recharge and discharge states, while the cbd shape has a significant impact on the libs discharge status only。
The prasher task group proposed a microflow model that included inter-particle glucose interaction and hydrodynamic interaction, predicting the viscosity of conductive carbon nanome and polymer binders and even the viscosity of the whole anode plasma, and found that the interaction between carbon nanoparticles depended to a large extent on the ratio of particles to polymer glues and the molecular mass of polymer glues, and that changes in particle interactions could be clearly reflected in the particle interassembling structure, thus reflecting the viscosity of the plasma。
Srivastava and others have illustrated the impact of am-cbd viscosity and intra-cbd fusion on the microstructure of electrochemical transmissions and related key properties (e. G. Ion curvature, electroconductivity and available am-electrolytic interface area), as shown in figure 2。
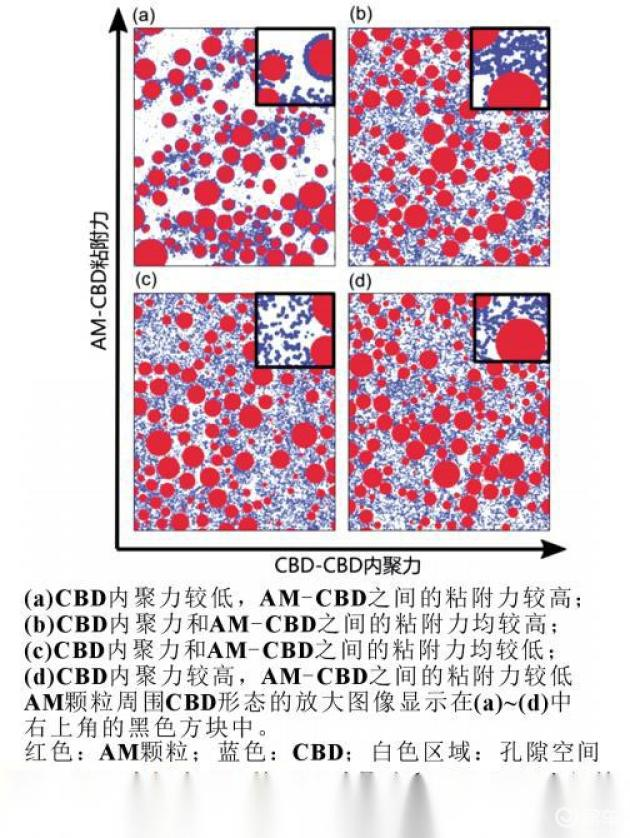
When internal concentrations are high, the concentration of the cbd around the am particles is significantly suppressed, and the cbd prioritizes the formation of grid-based gels and thus the availability of a larger am-electrolytic interface, thus explaining the reasons for the strong adhesiveness between the cbd and the am-cbd that significantly affects the properties associated with the electrochemical transmission of electrochemicals to electrodes, and based on this, a strategy to design the libs electro-chemical transmission properties of electro-chemicals by controlling the cbd and the bonding between the am particles and the cd provides a solution for optimizing the microstructure of the libs。
Common lithium ion cell electrodes gluer
2. 1 polyfluoroethylene (oil system)
Polyfluoroethylene (pvdf) is one of the first adhesives to be used, with high mechanical strength and wide electrochemical stabilization windows, and is widely used as a glue for various system electrodes, including libs. In the scale production of lithium ion batteries, high polar organic compounds such as nmp, dmf, etc. Are commonly used as solvents and pvdf is prepared as a soluble solution before being used as lithium cell glues。
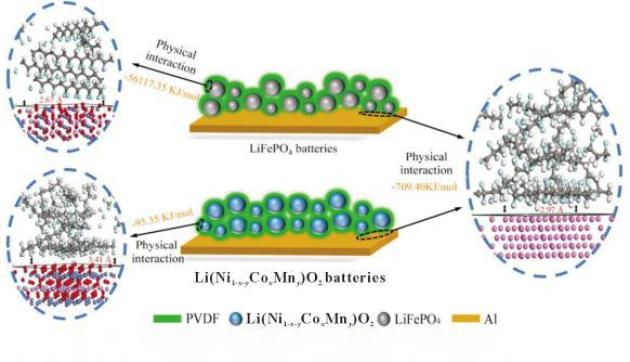
The combination of am and pvdf in libs was explored by zhong et al. Through the simulation of dft and analysis of the combination of am particles and glue in the libs electrodes (figure 3). Process simulations and theoretical calculations show that the combination of lfp and pvdf is much more than the combination of pvdf and al in lfp batteries, while in ncm batteries, the combination of ncm and pvdf is weaker than the combination of pvdf and al; analysis of scanning electron microscopes and the oie (aes) indicates that in lfp batteries, the main distribution of pvdf is on the lfp surface, indicating that pvdf is less adhesive in lfp batteries, while in pvdf batteries, both active materials and al surfaces are evenly distributed, indicating that pvdf has good visco properties in ncm batteries. The results of the study suggest that the development of new pvdf-based libs glues should focus on strengthening the combination between the glue and al and confirm that the interaction between am, al and pvdf in libs is primarily physical rather than chemical。
2. 2 pyromethyl cellulose and butzene rubber (water system)
Aluminium cellulose (cmc) is a cellulose linear polymer derivative formed by natural cellulose being replaced to varying degrees by aluminum. Cmc is a multifaceted weak acid that is able to detached from the formation of a cylinated root anional group, while its presence makes it easier for cmcs to dissolve in water than ethyl cellulose (ec), methyl cellulose (mc) and hydroethyl cellulose (hec), which enables cmcs to achieve water-based electrode production of solvents, which is more advantageous than pvdf in low-cost, non-toxic and harmless production. Cmc is considered to be a potential adhesive in the libs because it is capable of interacting with hydroxyl radicals on the surface of materials such as silicon/carbon and forming an ideal cbd network in electrodes, and because cmc is low-cost, hot-stable and clean of the environment。
Studies such as lee have shown that the suspension stability of graphite slurry, which replaces the lower cmc as a glue, is better, because the replacement of the lower cmc is more hydrophobic, increasing its interaction with the graphite surface in the water medium. Studies such as drofenik have shown that using a small number of cmcs (with a mass score of 2%) graphite negative poles can achieve the required effects of a large number of pvdf (10%) glues, and will not affect the normal decoupling of lithium ion in the graphite electrodes and the formation of the sei membrane, indicating that cmc use can reduce the use of glues, contribute to increasing the energy density of the libs electrodes, which is a good libs anode glue。
However, the cmc water glue has a strong rigidity and flaccidness, and the vacuum is dry, and the electrode surface of which cmc is a glue is clearly visible cracking, which may even lead to a gap between the electrode material coating and the fusion, resulting in the electrode “default”. In order to address this problem, liu, for example, is an elastic additive to cmc glue, comparing the effects of sbr-cmc composite glue to the traditional pvdf vis-à-vis the stability of the silicon (si) anode cycle, and exploring the mechanics and solubility of sbr-cmc composite glue in electrolyte solutions. The results show that the inclusion of sbr can effectively reduce the impurity of electrodes; the si anode using sbr-cmc composite glue shows a smaller volume of yang's modulus, a greater maximum extension, and a stronger attachment to the mc than the pvdf glue。
Research by the dahn team showed that the si electrodes, which were made from sbr-cmc composite glues, had a better capacity to maintain than the si electrodes, which were made from cmc glues only. At the same time, their studies revealed that, because cmc is a very hard and fragile polymer, cmc water glues work well as glues in electrodes with a high rate of particle size change in active substances. However, cmc water glue absorbs less organic carbonate electrolyte than pvdf, which may affect the multiplication of electrodes with cmc as a glue。
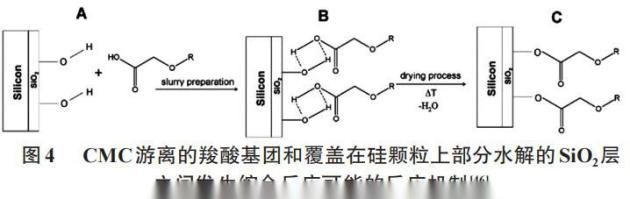
In addition, cmcs are used as additives to enhance the negative extreme cycling stability of libs (e. G., si and sn alloys), which show large volume changes during the battery cycle, and the mechanism for improved cycling performance is considered to be: (1) cmc chain bridge between si and carbon-conducted electron additive particles; and (2) cmcs create stable covalent keys (figure 4) or self-recoverable hydrogen keys on the surface of si particles。
2. 3 polypropylene acid glue (water system)
Polypropylene acid (paa) is a water soluble high-molecular polymer formed from monomer acrylic acids, which, because of their structural presence of large groups of acrylic acids (figure 5), have a strong interaction with active substances and aluminium aluminum and thus have better vistas, are potential high-performance glues to the libs electrodes, and during the libs cycle, pa can help to form a stable cei and improve the cyclic stability of libs。
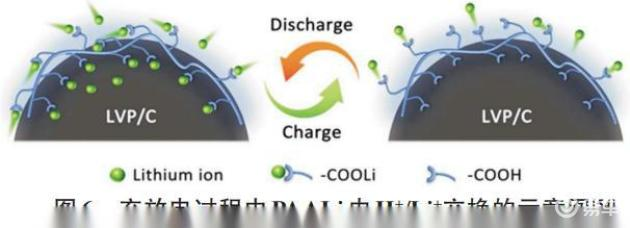
Su, among others, used paali for the first time as a visceral glue to li3v2(po4)3(lvp) and explored li+ transmission behaviour in paali and its effects on the electrochemical performance of libs. The results show that the new paaali adhesive displays excellent stability in organic electrolyte and has good adhesiveness with all electrodes, thus forming a continuous conductivity network in electrodes. The lvp battery using the new paali glue maintains 91% of its capacity under 10c after 1,400 cycles. Through test analyses such as ftir and x-ray electro-energy spectrum (xps), they have shown that paaali's glue can facilitate the transmission of li+ at the electropolar interface (figure 6), and that this electronic and li+ coordinated transport in paaali-lvp batteries enhances the electrodynamic properties of electrodes and provides a fast-track process for the reoxidation of the electrical capacity of the class, thus enabling them to obtain an excellent multiplier of 107 mah/g under 70c。
Chong, for example, studied semi-cell and full battery electrochemical properties of paax (x=h, l, na, or k) with sbr-added paas as a glue and graphite/lifepo4 battery systems with pvdf as a glue, showed that pvdf can effectively increase the initial coronal efficiency, reversible capacity and circulation stability of graphite/lifepo4 batteries compared to mpax (x=h, l, na, or k) and that a small number of (0. 5%~3. 0%) sbrs (sbrs) have avoided the generation of electrode dry and impurity cracks; in paax series, paaali and paana have better battery performance, which is attributed to their ability to produce more favourable polymer images (cei) in highly composite materials; and, at the same time, abax-series binders can reduce the manufacturing costs and environmental hazards of graphite/lipo4 batteries。
Summary and outlook
Although adhesives in the libs electrochemical non-active material, they are able to form the cbd structure in conjunction with the conductive carbon nanoparticles, and when the glue between the glue and the fusion is better, a good cbd conductive network can be formed by adjusting the viscosity between the cbd intensity and the am-cbd, not only allowing the electrode to have a strong mechanical and decomposition strength, but also enabling the electrode to form a conductive network that is conducive to electronic conductivity, making the electrode very efficient for electronic transmission; and contributing to an increase in the am-electrolyte interface for ion transfer, which reduces the degree of ion-transformation in the electrolyte, while also improving the quality of the passivation layers (e. G. Sei, cei membrane) formed on the surface of the am in contact with the electrolyte。
The main types of libs electrode glues currently used include the pvdf and the cmc, sbr, paa adhesives presented here. Pvdf has a good adhesiveness between the pvdf and the fusion, and can be modified by adjusting the polymerity of the fluoroethylene difluoroethylene (vdf) to its molecular mass, which is now widely used in electrodes production in various battery systems. Aqueous adhesives such as cmc, sbr, paa do not require organic solvents in practice compared to oil solubility vis-à-vis pvdf, avoiding contamination of the environment and damage to the health of operators by the vapour of high temperature organic solvents. Also, the cmc, a cellulose derivative with a wide range of sources and low cost, meets the low-cost requirements of the libs and has broad application prospects as an additive to improve the negative silicate extreme cycling stability; the paali glue is more adept and can complement the consumption of libs active lithium during the cycle, showing great potential for development and is expected to open up a new path to the development of libs high performance glues。