Response equation:
Cooh
Ii
Ii
Oh+ce
C-o-cch
Coo
Occh
Ii
Ch
Coo
Reactions:
Acetylene acetylene acetylene acetylene aque
The reaction temperature should be controlled at about 75 〜80°c and the following by-products may occur at excessive temperatures:
Live by-product: acetylene acetate aqueous acid acid
Oh°
Ii
C-o
Cooh
Cooh
Ococh
Cooh+ho
+h
Cooh
Aqueous acid
Ococh
Cooh
C-o
Ii
Acetyl acetyl acid
+h
Aspirin's rationale, steps and assembly
Purpose of the experiment
1 learn the principles and methods of the aylide response and master the aspirin preparation methods
2. Capturing methods to protect the oxidizing matrix
3 further mastery of crystallization techniques, installation and operation of filtering devices
Ii. The principles of the experiment
Aspirin is known as acetylic acid, which is white crystal, is soluble in ethanol, chloroform and acetylene, and is micro-solved in water. Factors
Heat, pain and inflammation treatment for flu, headache, fever, nervous pain, joint pain and rheumatism
It is also used to prevent cardiovascular diseases. Aa of the apc, which is commonly used as analgesic, is aspirin. The lab usually uses water
The acrylic acids and acrylic acids are produced by an aylide reaction under the catalytic effect of sulfate。
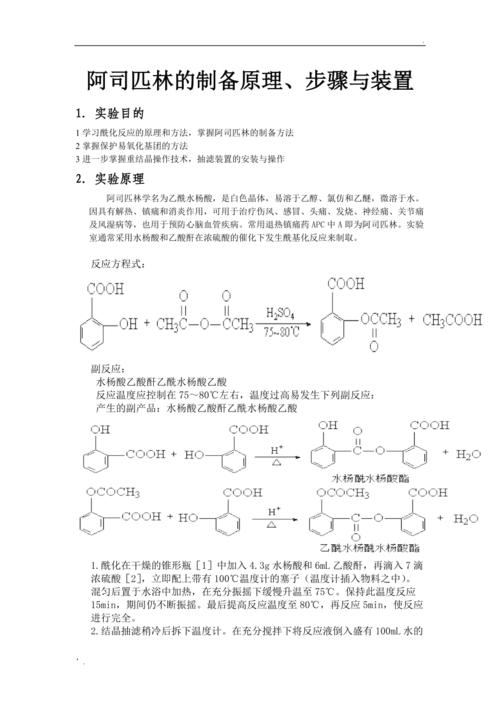
1-aylization in dry cone bottles [i] with 3g of cattle aqueous acid and 6 ml of acrylic acid and drops of 7 drops of sulphuric acid[2],
A plug with a 100 °c thermometer (thermometer inserted into the material) is immediately fitted. Heated in the water bath after mixing
Keep this temperature reaction slowly rising to 75°c under full vibration
A second-swipe water
Small amounts of impurities such as acetate water
Filter i
I-gee-seok
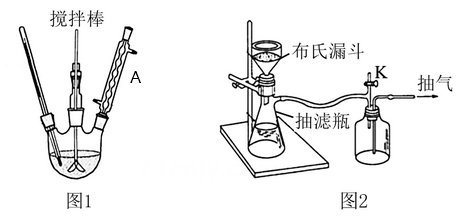
Iv. Experimental instruments
Round-floor flask (25ft1i), scales, glue drip tube, water bath, thermometer (1oo°c), glass bar, cloth
Sh funnels, vacuum pumps, cups (2ioml,iooml), reflow condensers, cone bottles (5oml or woml), test
Pipes, surfaces, cups, bath boilers
V. Experimental drugs
Aqueous acid (5), acetic acid dry mountains, sulphate (98 per cent), ice blocks, acetic acid heart, boiling stones, 1 per cent, 3 per cent
Iron chloride solution, ethanol, water (? 5%)
Attention
1acceleration reaction is performed by pressing the bottle plug with your hand in order to prevent a reaction of vapour rush. And it keeps shaking to ensure that the response is complete。
Control the aylide reaction temperature, otherwise by-product generation will be increased。
3 when the reaction fluid is transferred to water, it is to be fully broken and the large solid particles are to be crushed in such a way that it is not soluble when it is re-crystrated。
4-acetic acid is highly irritant and is to be used in the ventilation cabinet and be careful not to stick to the skin。
Additional note:
1 if aspirin is prepared in larger quantities, reflowers with electric mixers may be used. An electric mixer in the middle of the third neck bottle
Sphere condensers are installed at one side and thermometers are installed at the other。
The presence of hydrogen keys in the aqueous acid molecules hinders the phenolylization response. The reaction needs to be heated to five。
If a small amount of sulfate is added, it can damage the internal hydrogen key of the aqueous acid molecule, lowering the reaction temperature to about 8°c, thereby reducing by-products
Generate。
3 when dissolved, the heating time should not be too long or the temperature too high, otherwise the aspirin hydrolyzed。
Production rate calculations
Theoretical yield rate of actual production of products by appearance




