Common synthesis methods and comparisons of aspirin
Usually, aspirin uses acetic anhydride as an acetylizer for aqueous accelerides, while the catalysts chosen have a significant impact on the reprocessing, quality, productivity and cost of their synthetic products. The response was as follows:
Pilot principles for catalytic synthesis of trichloride soil:
Trichlorine is used as a luisic acid, which is soluble, filters filters, and washes with 5-10 ml (transferable solution, later). Combining and moving filters and detergents into 100 ml cups, slowly adding 15 ml 4mol/l hydrochloric acid. It's mixed with a lot of bubbles。
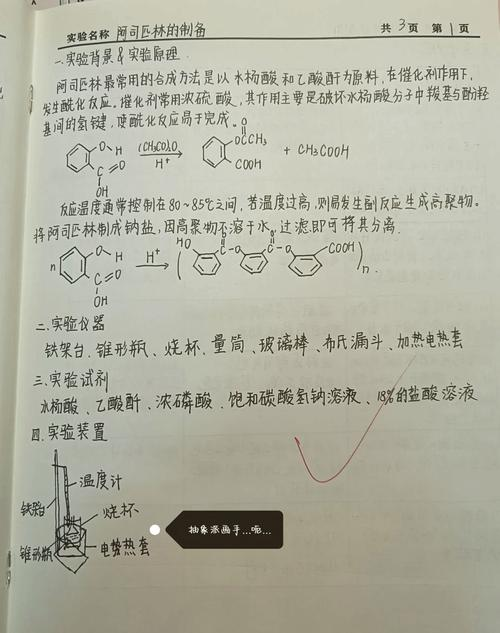
(3) filtration with ice water of 10m, 2-3ml cold water washing several times, drying. Dry. Scale。
(4) product purity test: take several crystals, plus 5 ml water and drop 1% fecl3 solution. Test purity。
Aspirin other synthesis methods:
Preparation process
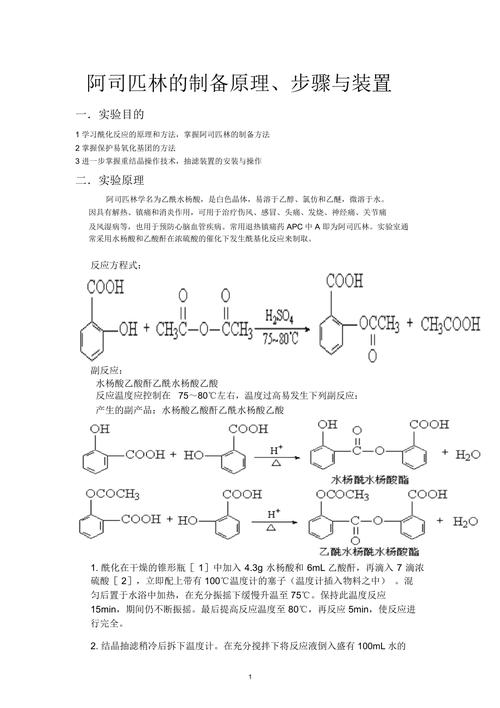
The mix is fed with flask with condensers, temperature is controlled at 150 - 160°c in the oil bath for approximately three hours, and acetic anhydride, which has been evaporated at reduced pressure, and the acetic acid produced in the reaction, has a weight of about 16 and a weight of 31 for the residual. Eighteen pures are available for re-crystaling with a double weight of benzene. If the concentration of residual fluids is increased, 10 pures can also be collected。
Preparation process
When phenolated acid is injected into the thorium, the temperature solubles, i. E. Cooling with icy refrigerants, i. E. Acetyl chloride, the material becomes slurry at the first drop, then liquid and then thicker. Heating on a water bath pot for 10 minutes, pouring on the ice and mixing to make the slime liquid solid, crushing the water and washing it and drying it up at 60 to 70 °c in about 13 products, can produce pure crystals in benzene。
Laboratories usually use the aqueous acid and acetic anhydride as a catalyst for the development of ayl fluoride response。
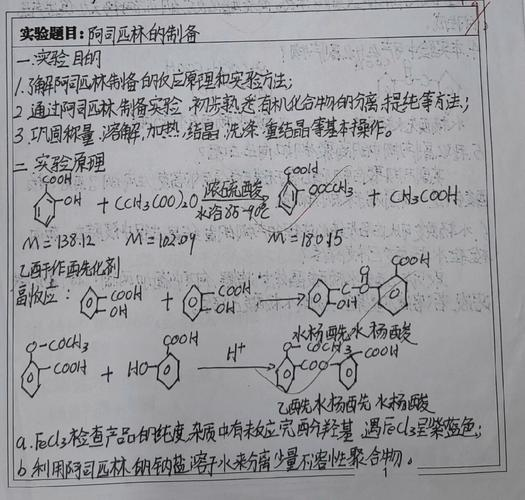
Other catalysts prepared for aspirin:
The synthesis of aspirin is based on the use of acetic anhydride as an acetylene as an accelerant and on the esterization of hydroxyl acid in water as a catalyst. The traditional synthetic aspirin catalyst, which is thick sulphate, has the following disadvantages. 1) low harvest rate (65% ~ 70%), corrosive equipment, acid drainage contamination. 2) operating conditions are stringent. Enriched sulphate is highly oxidized and reacts with strict control of its rate of accession and mixing, which otherwise results in the carbonation of the reaction. 3) when rough products are dry, partial oxidation of products due to incomplete sulphate separation leads to poor colouring of products. 4) the product cannot be heated and dryed, otherwise the remaining sulphate in the product can actuate acetyl acid to aqueous acid. The search for a new type of high-catalytic, environmentally friendly catalyst to replace synthetic acetylic acid acoustic acid has become a new subject of research. A comprehensive literature analysis shows that improved catalysts can be broadly classified as acid catalysts, alkalin catalysts and other types of catalysts。
The mechanism for acid catalysts to catalyse synthetic aspirin is as follows: acetic anhydride under acid acetic anhydride as a result of the electrostatic enhancement of the central gill carbon atoms centralyl fluoride is easily transferred to hydroxyl radicals to form esters, i. E. Complete synthesis of acetyl acid. The greater the acidity of the catalyst, the greater the mobility of the hydrogen proton, the easier it is to produce the ester, but the greater the acidity of the catalyst in the synthesis of the acetyl acid is also associated with the phenoloxydehydrate in the aqueous acid molecule and with another aqueous acid molecule, which produces more ester polymer by-products. As a result, based on the reaction of thiosulphate as a catalyst for the synthesis of aspirin, extensive research has been conducted on the substitution of thiosulphate as a catalyst for the synthesis of aspirin, with encouraging results. Acid catalysts include louisic acids, solid acids, organic acids, acid inorganic salts, acid inflated soil, etc。
Extractive soil is a non-metallic mineral resource with a two-dimensional route and a large hole molecular sift, with acidized acid-based inflatable esters. The greatest advantage of a chemicalized response is that it is high-receiving, and that the catalyst, when separated from the product by thermal filtering, can be used repeatedly, at low cost and without contamination of the environment, is a green catalyst that can be used to react。




