In recent days, the national drug authority has issued a bulletin on the revision of the chlorazine tablets (no. 64 of 2019) (hereinafter referred to as the proclamation), specifying that pregnant women are banned and that children under 12 years of age are unfit for use. In interviews with journalists, it was learned that chlorazine was in fact an atypical antipsychotic drug, which had been previously the cause of concern because of adverse reactions。

Atypical antipsychotics have caused concern
According to pharmacist zhang myung joo, the chief pharmacist of the shandong three colleges, chlorazine tablets are a psychopharmaceutical drug that applies mainly to various sub-types of acute and chronic schizophrenia, with a good effect on hallucinogenic and young. Emotional symptoms associated with schizophrenia (e. G. Depression, guilt, anxiety) can also be mitigated。
According to zhang myeongju, chlorazine is an atypical antipsychiatry, and the drugs listed in our country include lypone, agnostic, thallium, zirathone, arièle, zoropiron, paliketone and ammonium sulfur. Atypical antipsychiatry drugs are used mainly for the treatment of positive and negative symptoms of acute, chronic schizophrenia and various other psychiatric states. Atypical antipsychiatry is more resistant to mental illness than traditional antipsychiatrics and less conic reaction, and is therefore more widely applied to clinical purposes。
Previously, the adverse effects of atypical antipsychotics had raised concerns. In october 2015, the former national food and drug regulatory authority issued the drugs negative response information circular (no. 68), which focuses on the serious adverse effects of atypical antipsychiatrics, including chlorazine。
The adverse effects are mainly those of the central nervous system, such as dizziness, sleep addiction, insomnia, epilepsy, cone insular reaction, excretion, mania, pretence, etc. There are also serious adverse effects, such as particle cell deficiency and glucose metabolic anomalies。
The modified ban on pregnant women is not suitable for use by children under 12 years of age
The impact of atypical antipsychotics on patients ' blood sugar, blood resin and body weight is more well reported in the domestic literature and is clear in relevance. From 1 january 2004 to 30 june 2015, there were 2529 reported cases of atypical antipsychotic glucose metabolic anomalies, accounting for 3. 5 per cent of the total number of reported cases, including an increase in blood resin of 462 cases, an increase in blood sugar of 534 cases and an increase in body weight of 1533 cases。
In order to ensure the safety of the drug use, the national agency for drug control and drug control, in this revision, made recommendations for the use of the drug in a number of groups: there are not enough clinical studies of chlorazine among pregnant women. Pregnant women are banned and breast-feeding women should stop breastfeeding while using chlorazin。
In addition, the item [child medicine] has been revised to read that the safety and effectiveness of children using chlorazine is not clear. Children under 12 years of age are unfit for use。
It should be noted that, since a sufficient number of 65-year-old patients are not selected in the clinical study of chlorazin, it is not clear whether the response of older patients differs from that of younger patients。
The newly revised instructions should warn that the use of chlorazine therapy can result in low body blood pressure and an excess heart rate of about 25 per cent. Older patients, especially those with cardiovascular disorders, should be aware of the negative effects described above. Older patients are more sensitive to the anticholine effects of chlorazine, such as urine retention and constipation。
The dose selection of elderly patients is important and consideration should be given to the reduction of liver, kidney or heart function, complications or the sharing of other drugs. Other clinical trial reports indicate that older patients, especially older women, have the highest incidence of delayed physical activity disorders。
Zhang mingju said that, with the revision of the instructions, doctors would also be more cautious about the use of medication, while hospitals would be more stringent in their management and would have a positive effect on the safety of the use of drugs。
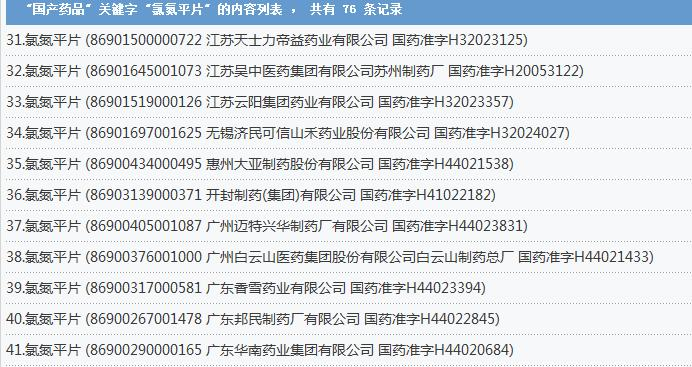
This is a revised version of the note, which covers many well-known pharmaceutical companies
Ziru's evening newspaper, ziru, a journalist, consulted the national bureau of medicine database and found that 76 national copies of the tablets were in possession of such well-known pharmaceutical companies as ziru pharmaceuticals, the shanghai shinyipine pharmaceutical industry, guangzhou city white cloud pharmaceuticals and jiang sunhua pharmaceuticals。
The market for psychostatics is large, and data show that the chlorine tablets sold 130 million in china's public health-care terminal in 2017. In industry analysis, the national drug authority requested a revised version of the tablet of chlorazine, which would have a small impact on a large number of pharmaceutical companies。
The national bureau of medicine and medicine requires that all chlorazine producers submit supplementary requests for amendments to the chlorazine tablets, in accordance with, inter alia, the relevant provisions of the drug registration and management scheme and in accordance with the request for amendments to the chlorazine tablets (see annex), to be submitted to the provincial drug control authorities by 12 october 2019。
Revisions concerning drug labelling should be accompanied by amendments; the instructions and other aspects of the label should be consistent with the original approval. All medical instructions and labels that have been released are replaced within six months of the filing of the supplementary application。
Chlorazine tablets producers should conduct an in-depth study of new mechanisms for the occurrence of adverse reactions, take effective measures to provide awareness-raising training on the use and safety of medicines, immediately notify drug operators and users in an appropriate manner about changes in content related to the safety of medicines, and guide physicians and pharmacists in the rational use of medicines。
Ziru evening, ziru




