Battery history
The history of battery inventions dates back to the end of the eighteenth century, when italian scientists, in 1780, while conducting a frog experiment, twitched the muscles of the frog's legs with a copper cutlery, as if they were stimulated by electricity。
Ford later considered this as a result of current stimulus between metal and frog leg tissue fluids。
It was designed by ford in 1800 as a device known as voltage reactor, with a negative pole for zinc and a positive pole for silver, using salt water as an electrolyte solution。
In 1836, daniel invented the world's first operational battery, the copper and zinc battery, and applied it to early rail lights。

Why is the battery so intransigent
The main measure of battery capacity is energy density, which is now, on average, only about 1 per cent of gasoline, which can be said to be quite poor。
Such a poor energy density is related to the material and structure of the battery, like our usual alkaline cell, which is manganese dioxide at the positive end, and zinc powder at the negative end, which is filled with electrolyte to transmit hydrogen ion, and when the battery works, the oxidation reaction between zinc and manganese dioxide produces 1. 5 volt volts of volts。
Looking at negative polar materials alone, zinc energy density is only about one tenth of gasoline, which can be said to be very low, whereas structurally, alkaline batteries are less than good。
U. S. B. Charge cell 5, alarm toy cell, charge battery
First, batteries cannot obtain oxygen directly from air as gasoline, and must provide for oxidizers and electrolytics。
The second battery also contains a large part of the containment material, such as, in order to prevent battery short circuits, all batteries must add a loop between two metal blocks, all of which significantly crowd out the available space for generating materials, dilute the energy density of batteries, low-energy battery materials and limited structural space, and constrain battery development。
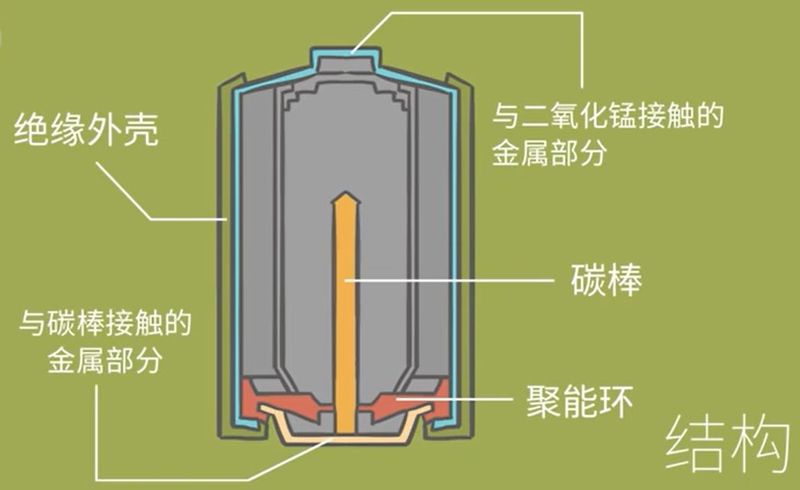
What's a fusion ring
Since all dry batteries are metal in their outer casings, they have to be separated by a positive-negative insulation to prevent short circuits and leaks, so that a plastic sheet is usually used to isolate the positive-negative poles, since plastic sheeting, i. E. Insulation, is the best option。
So this plastic sheet is designed to prevent short circuits and leaks, but some batteries do not see the plastic sheet because of the different negative and negative structures inside it, the plastic sheet is installed inside the battery, and others the insulated plastic sheet is placed outside, and this plastic sheet is a concentrated ring。
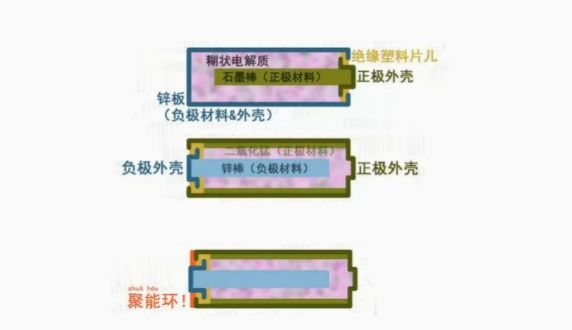
Battery works
Since the cell initially inserted two different pieces of metal in the salt water, the current has passed from one metal to the other through wires, and the pattern of battery work has remained unchanged for many years。
Dry batteries belong to the original cell in the chemical power source, with a metallic cap plus a zinc drum shell and conversion of chemical energy to electrical energy supply equipment。
For example, alkaline zinc manganese batteries, battery discharge is the electrolytic reaction of ammonium chloride and zinc, the released charge is a brass collector, the chemical reaction is oxidized because zinc is active, zinc is lost to electron oxidation, manganese is converted to electrons and zinc electrolysis is released from hydrogen gas, which increases resistance within the battery, and manganese dioxide is used to absorb hydrogen。
But if the batteries are working continuously or for too long, manganese dioxide becomes too late or is already saturated to absorb it again, the electrical resistance is too small for the output current to function, and if the batteries are heated or placed for a certain period of time, the concentrated hydrogen gas inside them will be heated out or slowly released to recover the manganese dioxide。

Battery manufacturing process
To see how the alkaline zinc manganese batteries commonly used outside are manufactured, the steel shell is first made into a 13 mm diameter cylinder。

The brass collectors in the batteries are the core of the batteries, cutting the brass wire to a length of 4 cm, and brass is a good conductor because of the high copper content, but the brass surface may also have oil stains, so that the cut brass will be rolled in the sand and the surface will be polished and the edges removed, and a nickel-plated cap will be welded on each brass。

Zinc will be easily released from the electronics and will be converted into paste through machines。

Compress the powder of the combined manganese dioxide graphite into a small ball form suitable for placement in the battery and then into a nickel-plating battery steel shell。

Batteries are assembled through vibrating discs and conveyor belts, re-inserted into alkaline electrolyte solution by the machine, with paste zinc added to the battery, with brass poles in place, each battery sealed by the machine, and the surface is packaged out of the plant after a packaging paper test。

As mentioned earlier, batteries are subject to structures and materials and are developing very slowly, but technology is constantly evolving, and now we see hope that a cell called permanent electricity is in existence, that electricity is always available and that nuclear power is being used? This battery cannot be used enough because it has been charged, and also how it has been used out。
The batteries are built into receivers to acquire electrical energy by accepting a radio signal delivered by an indoor transmitter, which, like wifi, is ubiquitous and does not affect human health. You can obtain continuous power supplementation wherever you are, like a remote control, and the clocks, which require batteries to be installed in electronic devices, no longer need to be replaced。

The creation of such batteries is likely not only to be end-users of alkaline and recharge batteries, but also to reduce environmental hazards, as there is no need to replace them, thus reducing the disposal of used batteries, which is expected to be applied at an early date。