We often hear or see traffic accidents as a result of drivers' drunk driving, and in the news reports of accidents, we often say what alcohol is in the driver's blood and what is the legal limit for alcohol in the blood. For example, a driver may be tested for alcohol at 0. 15 in blood, with a statutory ceiling of 0. 08. But what do these figures mean? How can the police determine whether a driver suspected of driving alcohol is legally ingestion? You may have heard of the snorting alcohol test, but you'd like to know how a person's gas is showing his alcohol intake。
In the light of public safety, it is important to prohibit drivers from driving after drinking. In 1999, 42 million people died in traffic accidents in the united states, 38 per cent of them alcohol-related. Even for drivers who are able to pass sober tests by touching their nose or walking straight lines, alcohol intake may still exceed the statutory limit of blood alcohol content and become road killers. As a result, the police used some of the latest techniques to detect alcohol concentrations of suspected drivers and to take them off the road。
Many of the duty traffic police officers use excretion alcohol testing equipment (one of which is the excavator) to determine the blood alcohol concentration (bac) of the driving suspect. In this paper, we will study the scientific principles and techniques that are present in these pneumatic alcohol testing devices。
1. Why test
The legal basis for driving alcohol is the blood alcohol concentration (bac) level. However, after taking blood samples at the scene, laboratory analysis is neither practical nor efficient for the detention of drivers suspected of driving drunk (dwi) or driving drunk (dui). The urine alcohol testing proved to be as impractical as blood samples. What we need is a way of measuring indicators related to blood alcohol levels without intrusion into the driver's body。
In the 1940s, for the first time, a gas-gas alcohol test device was invented for use by the police. In 1954, dr. Robert borkenstein of the indiana police department of the united states invented the exhale alcohol tester, which is still used by law enforcement agencies。
Let's see what these devices are based on。
2. Test rationale
The alcohol that a person drinks is absorbed into his or her blood by mouth, throat, stomach and intestinal tracts and exhales in the form of breath。
Alcohol is neither digested nor chemically altered in blood after ingestion. Because alcohol is volatile, it can evaporate from the blood when it flows through the lungs, so some alcohol enters the gas in the lung bubble through the pulmonary filament. Alcohol concentrations in pneumatic gases are associated with blood alcohol concentrations, which can be tested through pneumatic alcohol testing devices when the alcohol in pneumatic gases is out. The traffic police do not have to extract the driver's blood to test their alcohol levels, but rather test the driver's breathing on the spot so that they can know immediately whether the driver belongs to a drunk driver。
Because the alcohol concentration in the exhalation is related to the alcohol concentration in the blood, the traffic police can calculate bac by measuring the alcohol content in the exhalation. The proportion of alcohol in breath and blood is 2100:1 (translation: original). This means that 2100 ml of pulmonary bubble air contains the same alcohol as 1 ml of blood。
According to the american medical association, when blood alcohol levels reach 0. 05, the body is damaged. If a person's blood alcohol concentration is measured at 0. 08, this means that 0. 08 g (80 mg) alcohol is present per 100 ml of blood. Over the years, we have implemented a drinking driving standard of 80 mg alcohol per 100 ml of blood, to be replaced by 0. 08 below。
The traffic police now have several different devices to measure bac。
Exhalation analyser
There are three main types of pneumatic alcohol analysis equipment based on different principles:
In either type, each equipment has a blow-out vent, a pipe for drivers to blow, and a sample room for air storage. The rest of the equipment varies according to type。
Exhale alcohol tester
Excretion alcohol testers include:
To measure alcohol, the driver breathes into the instrument. A sample of the gas produced reacts in a vial with a mixture of sulphuric acid, potassium chromite, silver nitrate and water. The principle of measurement is based on the following chemical reactions:
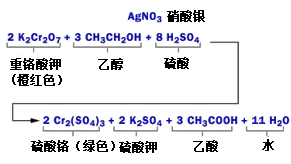
In this response:
Sulphuric acid dissolves alcohol in air into liquid solutions。
Ethanol and potassium chromiumate reaction generation: chromium sulfate, potassium sulfate, acetic acid, water
Silver nitrate is a catalyst that accelerates the reaction without involvement in chemical reactions. In addition to transferring alcohol from the gas, sulphuric acid may provide the acid conditions required for the response。
In this response, red orange heavy chromate ion becomes green chromium ion when the ethanol reacts; the degree of colour change is directly related to the amount of alcohol released from the air. In order to determine the alcohol content in air, the reaction mixture is compared with an unreactive mixture in the pv system, generating currents and moving needles from static positions on the instrument. The operator then rotates the knobs and takes the needle back to its original position, and the more the knobs rotate, the higher the alcohol content, and thus the alcohol content。
Chemical composition of alcohol
Alcohol in alcoholic beverages is ethanol. The molecular structure of ethanol is as follows:
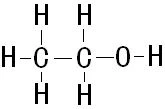
And the c is carbon, h is hydrogen, o is oxygen, and each connector is the chemical key between the atoms。
Molecular hydroxyl radicals (o-h) are official groups of alcohol. There are four keys to this molecule:
Atom chemistry is a shared electronic pair. Chemical keys are like springs: they can bend and stretch. These properties are important for ethanol in infrared spectroscopy samples。
4. Exhalerators
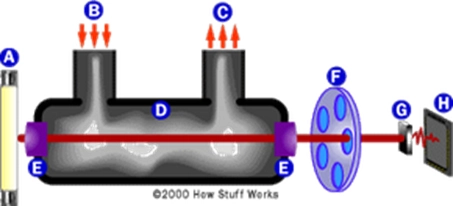
Exhalation intoxicator diagram. A quartz lamp (infrared light source), b blow-out entrance, c exhalation exit, d sample room, e lens, f filter wheel, g photovoltaic tube, h microprocessor
This equipment uses infrared spectroscopy to identify molecules based on infrared light absorbed by molecules。
Molecular vibrations continue and change when molecules absorb infrared light. Vibration changes include the bending and stretching of various keys. Each key within the molecule absorbs infrared radiation of different wavelengths. Therefore, in order to identify the ethanol in the sample, you must observe the wavelength of the chemical keys in the ethanol (c-o, o-h, c-h, c-c) and measure the absorption of infrared light. Absorption wavelengths can identify the substance ethanol, and the amount of infrared absorption tells you alcohol levels。
In the excavator:
5. Alcohol sensors iii or iv
Modern fuel cell technology has been applied to respiratory alcohol detectors. Fuel cells for equipment such as alcohol sensors iii and iv。
The fuel cell has two platinum electrodes, which contain the electrolyte material for polygonic acid in the middle. When the gas from the suspect passes through the side of the fuel cell, the platinum produces all the alcohol in the gas oxide, producing acetic acid, proton and electronics. The electrons flow from platinum electrodes across the guidance lines. The route is connected to the current meter and the platinum pole at the other end. Protons pass through the lower part of the fuel cell and combine with oxygen and the other side electronically to form water. The more oxidized alcohol, the greater the current. Microprocessors measure currents and calculate bacs。
Operators of pneumatic alcohol testing devices must be trained in equipment use and calibration, especially if the test results will be used as evidence for drinking driving tests. Law enforcement officers may carry portable respiratory testing equipment in the same way as full-scale equipment. However, the judge's decision may be based on the accuracy of the respiratory test, and the prosecutor is therefore more inclined to obtain results from full-sized equipment。
Alcohol oxidation
If, in the presence of oxygen, hydrogen is dropped from the carbon to the right of ethanol, acetic acid is obtained, which is the main ingredient of vinegar. The molecular structure of acetic acid is as follows:
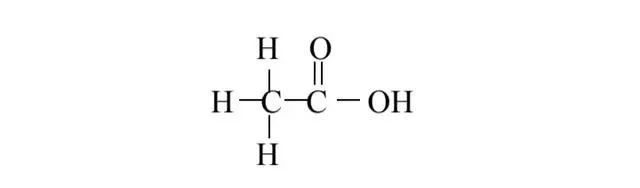
In which c is carbon, h is hydrogen, o is oxygen, the connector is a single key between the atoms, the symbol "=" is a double key between the atoms. When ethanol oxides to acetic acid, two protons and two electrons are also produced。
By craig freudenrich
Translation: deep
Reviser: the scavenger
Original link: how breathalyzers work