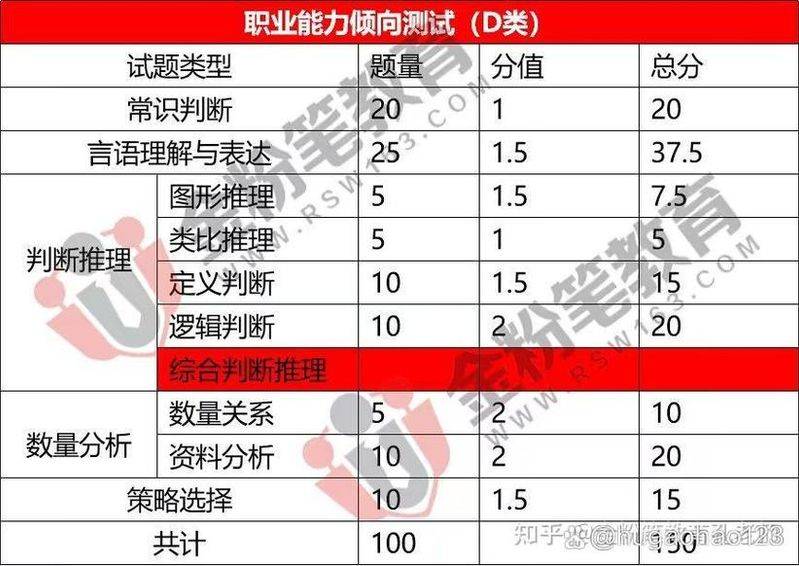
1. Fire safety training at school, fire safety training at school, i. Fire safety basics, fire safety basics, concept of combustion: burning is thermal reaction of flammables and oxidants, usually accompanied by flames, light and/or smoke. (i) fire is defined as a burning that is out of control in time and space. (ii) category a fire: a solid substance fire. This substance is often organic in nature and generally produces thermal residuals during combustion. For example, timber, cotton, wool, plum, paper fires, etc. Category b fire: a liquid fire and a melting solid fire. Examples include gasoline, kerosene, crude oil, methanol, ethanol, asphalt, paraffin fires, etc. Category c fire: refers to gas fire. Examples include gas, gas, methane, ethane, propane, hydrogen fires, etc. Category d fire: means metal fire2, such as potassium, sodium, magnesium, titanium, zirconium, lithium, aluminium magnesium alloy fires. Category e fire: electric fire. A fire with electrical objects. Fire protection basics, (iii) general reasons for fire, (iii) general reasons for fire, (i) general reasons for fire, (i) human factors (e. G., setting fire, throwing cigarette butts, breaking fire, etc.), causes of equipment (e. G., overloading of electricity, lack of maintenance, etc.), causes of materials (e. G., combustible materials), causes of the environment (e. G., lightning, self-burning), causes of management, basic reasons for fire protection, and causes of fire at school, in particular: (i) imprudent use of light fire, causing fire fire fires, violation of the protocol, basic fire safety principles, (iv) strengthening fire safety management, (iii) strict control of fire sources, adoption of fire resistant buildings, prevention of the spread of fire, limitation of the extent of possible development of fire, organization of fire-fighting units, provision of appropriate fire-fighting equipment. The basic common sense of fire suppression, i. E. The basic method of fire suppression destroys the already established conditions of combustion, so that the combustion can be extinguished and the fire hazards minimized. According to the principles of combustion and the operation of fire suppression operations, the basic methods of fire suppression are 1: cooling. Asphyxiation act. One, cooling method. It is the direct injection of fire extinguishers into the incendiary material to reduce the temperature of the incendiary material below the flame, to stop the combustion or to spray the fire extinguisher on objects near the source of the fire, free from the threat of fire heat and to avoid creating new fires. (water is a fire extinguisher with a very cooling effect). Asphyxiation. It is to prevent the flow of air into the burning zone or to dilute the air with non-fired substances, so that the incendiary material does not have enough oxygen to extinguish it. (the main effect of foam fire suppression is asphyxiation). 3. Separation act. It is to isolate or remove the flammable material from the source of the fire or around it, and the combustion ceases as the flammable material is isolated. That's how it's done. 4. Depression laws. It is the involvement of the fire extinguisher in the course of the combustion reaction, the disappearance of the free radicals generated during the combustion, the formation of a stable molecule or active free radical, and the termination of the combustion reaction. (halogenated alkane extinguishers and dry powders inhibit fire suppression). Fire suppression basics, (ii) fire extinguisher type dry-foamed halogenated carbon dioxide fm200 (heptafluoropropane), fire suppression basics, (iii) different types of fire extinguisher selected to save fire type a: water type, foam, ammonium phosphate dry powder, halo-alkane fire extinguisher selected; fire type b: dry powder, foam, halogenated alkane, carbon dioxide fire extinguisher selected; fire type c: dry powder, halogen5, alkane, carbon dioxide fire extinguisher selected; fire type d: should be addressed by design and local laboratory fire control. (b) basics of common sense for fire suppression, (iv) method of use of fire extinguishers, and removal of safety barriers. 2. Point the piping towards the fire. 3. Press the handle hard, select an upper wind position close to the fire, and shoot dry powder into the flame base. 4. The principle of allocation of fire extinguishers after extinguishing and cooling smoke (v) with water should not be less than two in a fire extinguisher site. It is not appropriate to have more than five fire extinguishers per location. Fire suppression basics, (vi) other fire suppression equipment, 1 system, outdoor flammation system, 2, indoor flammation system, 3, automatic fire alarm system, 4, automatic water spraying system, 5, foam fire suppression system, 6, gas fire suppression system. Fire escape knowledge, fire burning products, all substances produced by combustion or heat, decomposition, are called combustion products. It usually refers to the gas, heat and visible smoke generated by combustion. Fire hazards to victims are comprehensive, with four main kinds of hazards to people at fire sites: oxygen deficiency, high temperature, toxic gases, dust, and the combined effects of smoke, high temperature, and humans. Fire escape knowledge, i. E. Smoke hazard and protection, carbon dioxide, carbon monoxide and oxygen content in air: normally: 0. 06% co2 and approximately 21% oxygen. In case of fire: co2 up to more than 13%; carbon monoxide up to 1-2%; oxygen content down to 19-16% when fire is high: oxygen content down to 7-6%. On 5 june 2009, at approximately 0825 hours, 25 people were burned to death and 76 were burned in a bus near the three circles in cheng city. There was a fire on the public transport vehicle 6. 7 bus at the central television station north, located at 7 north and around 20:30 p. M. On the north annex to the city of kyoto, and on the north side of the main view building, the “oriental hotel of beijing”. The hotel building was about 159 metres tall, and there was a fire on both the east and the south, which was over 100 metres high. Following a call from beijing city command centre 119, 16 squadrons and 54 fire trucks were quickly deployed to the scene to view the fire at the new site as a result of the firm's insistence on burning fireworks in category a. The fire annex is not yet operational and only a small number of personnel remain on duty. The fire in the ocvi building (hotels, studios, etc.) was 100 metres high, there was a fire scene, traffic was blocked on the ground, the fire department used high-pressure water guns to control the fire, and there had been no effective precipitation in beijing for 108 days and air drying had risen. Air moisture in the evening of 9 days is around 50 per cent and remains relatively dry. Zhang jianyong, a brave victim of death, is 30 years old and is the director of the red temple squadron of the fire brigade. In addition to his heroic command and his dedication to the fire, he removed his own gas mask from the scene in an emergency situation to the personnel he needed. High school kids played games that led to a fire, two people were burned, fire water was extinguished, the wounded were taken to hospital, four students spent christmas eve together, and they stayed late because their parents weren't at home, and the game power was not shut down, and the machine overheated caused a fire. Two people were killed, one escaped and one was rescued. All four students were students in the second and sixth grades of beijing 159 high school. There was a fire in the 3rd building, room 601, of the 3rd block, 3th wing, of the street road where they lived. One of the boys climbed out of the window and one of the boys climbed to the roof and was rescued by firemen with a ladder. Two other students died as a result of suffocation. Firefighters, in the course of their rescue operations, delayed the rescue, as many private vehicles were parked on the road leading to the scene. The fire burned for about two hours. (05. 12. 26)9 on 7 september 08, at around 22 p. M., there was a fire at the chemical plant of the 21st high school in the gate of kaguyama city, municipality of fukuzhou, in which dozens of tons of chemical materials were piled up, which quickly spread to the chemical depots and could explode at any time, endangering the lives and property of the surrounding population. More than 120 troops from the four fuzhou detachments of 17 fire trucks arrived at the scene and were successfully extinguished after more than two hours of fire, resulting in no casualties. Two girls were killed, dozens of tons, as a result of the 2. 2 fire in chai tong, lake city, which resulted in the death of two 6-year-olds (playing a lighter) and a 2. 23 fire in xiamen city, located below the ridge of lake city. Methamphetamine threatens the surrounding villages. Fire protection immediately evacuates thousands of people, 20 tons of foam covers fire suppression, toxic gases fumes several firefighters, fire escape knowledge, 1. Levels of carbon dioxide, carbon monoxide and oxygen in the air and human function 10 are: breathing difficulties, dizziness, cough when carbon dioxide in the air reaches 2 per cent; life is at risk when more than 5 per cent; and when 20 per cent is reached, death can occur within a short period of time. When the carbon monoxide content in air reaches 1. 3 per cent, humans are intoxicated. When oxygen levels fall to 10 per cent in the air, people find it very difficult to breathe; if they fall to 6 per cent, they immediately choke to death. Fire escape knowledge, with carbon dioxide concentrations at the top of the room in 11-13 minutes after the fire being tested at about 9 per cent; in the centre at about 5 per cent; and on the ground at about 2 per cent. Carbon monoxide is lighter than carbon dioxide, most of which is concentrated in the centre of the room, equivalent to about 0. 8 per cent of human breathing。as a result, less carbon dioxide and carbon monoxide were inhaled in the position of the smoked rooms down or down. Fire escape knowledge, fire escape knowledge, 11, 3, if burned with plastics, fibres, etc., generate additional gases such as photogas (carbonyl chloride cocl2), chlorine gas, and hydrogen cyanide, which, although of limited concentration, are extremely hazardous. When chlorine is at 0. 1, the inhalation results in osteoporosis and severe eye damage, as well as pneumonia, emphysema and pulmonary haemorrhage; when the concentration exceeds 2. 5, it causes immediate asphyxiation. Hydrogen cyanide levels of 0. 027 and photogas levels of 0. 005 can immediately kill people. Cyanide is a compound of nitrogen, carbon, non-coloural gases, highly toxic, which causes acute poisoning when humans are inhaled in large quantities of cyanide, and the heavy ones are immediately unconscious and their breathing is stopped within 12 minutes; the light ones are first fainting, unconscious, vomiting, then heart attack, difficulty in breathing, convulsion, coma, and last breath is stopped. Fire escape knowledge, smoke protection at the fire scene can be done by preventing smoke from entering the room, closing doors and windows that are connected to the burning place, blocking the gap between doors and windows with water-drinking clothes, and covering with wet towels. Shut your nose to reduce smoke inhalation; don't shout; find the right place to avoid smoke temporarily; and, fire escape knowledge, (ii) how to escape fire often leads to the following behaviours: habit of lighting, fear of fire, narrow survival, blind escape from building and fire escape knowledge, what to do when you're caught in a fire? 1. The first consideration is where the fire was fired. The direction of escape is determined by identifying the fire, looking at the location of the flame or the fire, identifying the direction of the black smoke or air flow. Simple way of determining: a, where the black smoke comes from, or where the air flows along the ground is the point of fire. B with a flashlight, black smoke flows in the direction of the fire. C. With two fingers, with spit on it, the party with a cooler finger is the source of fresh air. Fire13, escape knowledge,2 during flight, wet towels were used to cover the face, blankets were used to prevent or reduce inhalation of toxic fumes and to move forward. 3. If the fire has burned to their own room and cannot escape, they have to flee from the window down, find a rope or tear the sheet open and grow a strip, one of which is fixed on the door and window and slides down or down the water pipe. If the length of the rope is not sufficient to reach the ground, it is possible to escape by kicking glass into a safe room as long as the rope is used to reach the floor where there is no fire. The fire escapes knowledge, fire escapes knowledge, and if the fire is already high when you find it. 1. If the smoke is so strong that only the sound of the ambulanceman is heard and the ambulanceman does not know where you are, you shall knock on the hose or the heating pipe, and use your handlight to shine outside, and you may also reach out the window and throw something out to get the fireman's attention. 2. In case of fire, 14 shall make every effort to leave the fire zone quickly and not to hide, especially under the bed, table or closet, so that even if the fire does not burn, it will be poisoned by smoke and you will probably not be found by the firefighters who come to rescue. When you flee the scene of the fire, you must not use the elevator to avoid being trapped in it. 4. Before opening any door, you must touch it with your hand. If the doors are hot or smoked from the doors, find another way. Even if it's cold, you have to be careful, because when you open the door, you bring in oxygen and rekindle the mars inside. 5. When a safe passage is blocked by the flames, the old and the sick shall close the doors and windows and place wet sheets on the sutures of the doors and pour water on the doors in order to prevent the smoke and fire intrusions and wait for help. Fire escapes knowledge, and if your route is a sea of fire and you have no choice, climb along the wall 15 and pass. Your clothes are on fire, and stop and roll on the floor. If someone is on fire and is not warned in fear, he should be knocked down as if he had been rescued in the water, then he should roll until the fire on his clothes has gone out. Fire escape knowledge, the following methods have been helpful in escaping fire: one, a list of public exports. It is not for you to know where there will be a fire. 2. Ensure that each bedroom has two exits. This means that when it is not possible to escape from the door, it can escape from the window or the balcony. It ensures that every family member, especially children, is able to climb up the window stage by opening windows. 3. Higher-level dwellings should be equipped with ropes. And make sure it's found in time. A fire-fighting water belt in a fire-fighting window can be used as a means of escape. Five exercises should be conducted twice a year. 16. It is only through repeated repetition that the fastest possible escape from the fire can be guaranteed. 6. Do not waste time on clothing and stealing valuables, knowing that life matters more than anything. The basic common sense of fire suppression, the doctrine of water fire suppression, absorbs 1,000 kilocalories of heat at 1 kg of water. The total evaporation of 1 kg of water requires the absorption of 539. 9 kilocalories. When water is exposed to hot incendiary incendiary matter, vapour produces a large amount of water vapour, prevents air from entering the burning area and dilutes oxygen levels in the burning area, which reduces the intensity of the burning by a gradual shortage of accommodative oxygen. Fire suppression basics, fire suppression principles: fire suppression foams form numerous small bubbles by chemically and physically filling large amounts of gas (co2 or air) with water solutions from foam extinguishers. Its weight is much smaller than that of generally flammable, flammable liquids, which can float to the surface of the liquid and form a foam cover. Fire extinguisher foams are also 117 incorrigible and can be attached to the surface of generally flammable solids. (b) fire suppression basics, fire suppression principles for dry powder: dry powder extinguishers are stored in dry powder fire extinguishers or dry powder fire extinguishers, and the amount of fire extinguished by pressure on the pressurized gas (co2 or nitrogen) ejected the dry powder from the nozzle to form a mist stream of gas-pressurized powder that is fired at the burner. When dry powder is in contact with the flame, a series of physico-chemical effects occur and the fire is extinguished. Dry powder is divided into two main categories according to the range of use: general dry powder extinguishers: sodium carbonate; more dry powder extinguishers: ammonium phosphate; basics of fire suppression; and fire suppression principles for halogenated alkanes: halo-alkyl fire extinguishers are interrupted by inhibiting the chemical reaction process of combustion, which serves the purpose of fire suppression and is performed by removing the active substance in the combustion reaction, known as the chain break or inhibition process. Classification: 1211 (cfc-1-bromomethane) 1301 (cfc-methane) 118, 202 (cfc-2202) 2402 (tcf-113) note: due to the release of this fire extinguisher, storage in the atmosphere for up to 87-110 years would have a significant adverse impact on the environment, and in order to protect the ozone layer, the use of halon-type water extinguishers would be banned globally in 2005 under the montreal international convention organization. (b) basic knowledge of fire suppression, co2 fire suppression principles: absorb large amounts of heat (approximately 138 kilocalories per kilogram of liquid co2 gasification). It has some cooling effect on the burner. Increases in the composition of the air, i. E., does not burn or contribute to combustion, and reduces the oxygen content of the air in relative terms. When carbon dioxide reaches 30-35 per cent of the air, it can gradually suffocate the general flammable material to 43. 6 per cent, inhibiting the explosion of gasoline, vapour and other flammable gases. In general, fire suppression, fm200 (hexafluoropropane) is superior to fire extinguisher: a mixed gas containing nitrogen, carbon dioxide, argon, a fire extinguisher with high blast resistance. It has characteristics that do not conduct electricity, do not damage equipment, and are extremely toxic to humans. The fire-fighting concentration of the fire extinguisher was 5. 8 per cent, which was below the critical mass concentration levels that had been tested for adverse human effects. Undestructed to the surrounding environment after release, short atmospheric residual time (31-42 years) and low relative fire-fighting concentrations。i don't know,