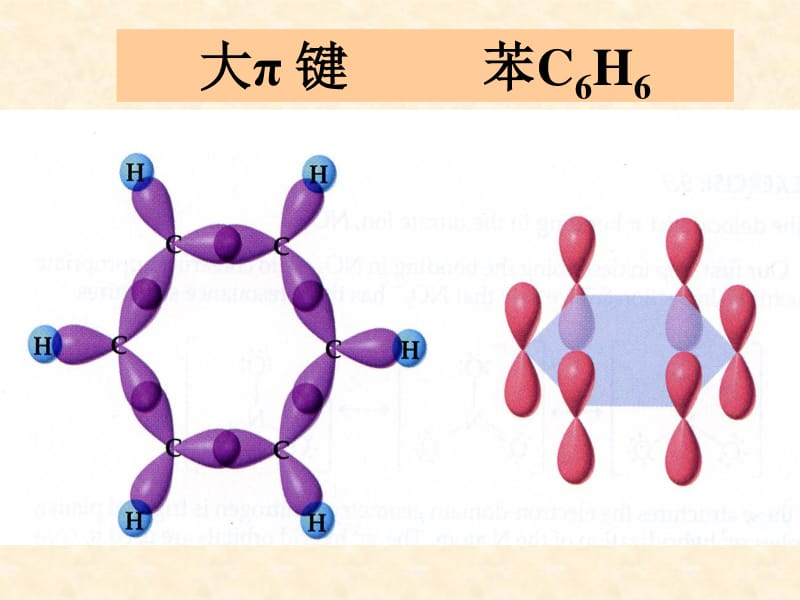
[synthetic] modified orbits. Because each orbit contains one fourth of the s-orbital composition and three quarters of the p-orbital composition, we call this orbit a sp3 misinformation orbit. Mixing orbits are used to accommodate key electrons and stand-alone electron impregnations: ch4 molecule formation 2s 2p 2s 2p 2p in order to minimize the exclusion between orbits in space, with the extension of four mixing orbits pointing to the four vertices of the positive quadrilateral, respectively. Mixing orbits: a new set of orbits formed by a combination of atom orbits. The four c-h single keys of methane should be considered, yet the four price layer atoms of carbon are three vertical 2p orbits and one spherical 2s orbits that overlap with the 1s atom orbits of four hydrogen atoms, and it is impossible to get a tetrahedron-shaped methane molecule. Based on the principles of electronics, the following groups of molecules are judged to belong to the same electrons: a. H2o, h2sb. Hf, nh3c. Co, co2d. No so2a, in substances consisting of short-term elements, and no2-equivalent electrons are:, the electronics are similar in nature. The chemically-transforming formula of the electron principle equals the total price of the electron and the molecule or ion has the same structural characteristics. Molecular or ion equivalents of electrons that meet the principle of electronic equivalence. Some of the applications of electronics: (1) determine the stereotyping of some simple molecules or ions: the electronics generally have the same stereotyping applications (2) for the manufacture of new materials. Hhe li be b n o f na mg al si c cl ar o3so2 mixed orbital theory section ii covalent keys and molecular stereotypes i and some typical molecules 176. Why is methane a orthodox? In order to resolve this contradiction, pauline proposes the theory of modicum orbits -- thinking about carbon atoms electrons: 2s22p2 atoms atoms atoms. Mixing orbital type: impregnated results such as sp sp sp3: redistribution of energy and spatial direction, equal in number of keys