Figure 2. 3 fluid bed gas phase catalyzing with hydrogen benzidine process figure
(2) nitrobenzene liquid-catalytic hydrogen process
Nitrobenzene liquid phase-catalyzed hydrogen, industrial production units typically use high-activity precious metal suspension catalysts, and sludge and fluid bed reactors are commonly used, and liquid phase-added hydrogen reactors tend to have a greater production capacity than homogenesis. The hydraulic phase is pressurized at 80 - 250°c, and the rate of single-way transformation of nitrobenzene is usually 98 - 99%, and benzidine is refined by decompressive distillation. The process process of the hydrobiphenyl phase-up process is shown in figure 2. 4。
Loading...
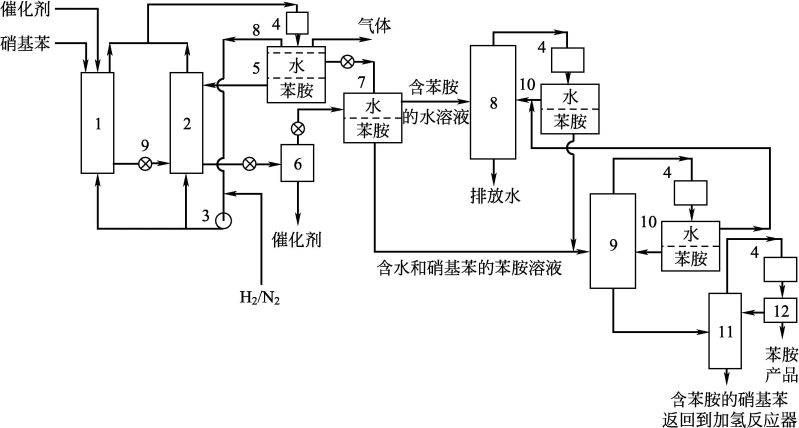
Figure 2. 4 ici nitro-benzene lcd process figure
1,2-helium-added reactor; 3-gas cycling pump; 4-condensers; 5-initial separators; 6-filter filters; 7-second separators; 8-water distillation units; 9-benzidine distillers; 10-separators; 11-benzidine distillation towers; 12-reverts device
Ici developed continuous liquid catalytic hydrogen processing in the 1960s. Benzamine as a solvent, hydrogen temperature close to the solvent boiling point, pressure of less than 100 kpa, reaction heat partially or entirely absorbed by evaporation from the product, and a large volume of amphetamine back into the reactor to maintain stability in operating conditions. Using nickel/silicate earth catalysts, the particle size of the carrier is 200 oculars。
Du pont uses precious metal catalysts, which are highly active and have a long life and do not lead to benzene cycling and hydrogen. Dupon uses a carbon-borne platinum/polymer: pd (4. 5 per cent)-pt (0. 54 per cent)-fe (5 per cent)/c for iron as a degenerative agent. The use of a modified agent can prolong the service life of the catalyst and increase its activity, which is reflected in a piston mobile bed reactor。
In order to overcome the high levels of impurities in the ici nitrobenzene response system, mihai east asian chemical series in japan uses the method of reducing the concentration of nitrobenzene in the reaction to increase the purity of benzidine, using precious metals pd, pt/c catalysts, assisted by sodium carbonate and sodium carbonate as catalysts, and responding to the response system by adding zinc compounds, 150 ~250°c and 0. 3 ~0. 7 mpa without water. The concentration of nitrobenzene in the reactor is maintained at 0. 01% or lower, with continuous evaporation in the form of vapour of the products phenylamide and water. The technology has been more effective in inhibiting the production of substances containing hydrogenized nuclear materials, providing access to benzidine, which is largely free of non-reactive nitrobenzene, and adding large quantities of solvents to control the reaction temperature through solvent subthermal。
The advantages of lhp are low reaction temperature, low side-response, high catalyst load, long life and high equipment capacity. A device producing 100,000 tons of benzidine a year, using gas phase, requires several large-sized reactor sets, while a liquid phase, with less than half the size of gas phase, requires complex inner packaging. The liquid phase method does not use large-cycle hydrogen gas cycles, while the water cycle uses pumps instead of hydrogen-cycle compressors. Shortcomings are that the reaction must be separated from the catalyst, the reaction and the solvent, and the operational maintenance of the equipment is costly。
2. 1. 1. 3 nitrobenzene plus hydrogen benzidine catalysts
Nitrobenzene and hydrogen benzidine catalysts can be classified as copper, nickel and precious metal catalysts。
(1) copper catalyst
Copper is a catalyst material that is easy to obtain, simple to prepare, low-cost, selective, but less toxic, with trace sulphides highly toxic to catalysts. The cu/sio2 series consists mainly of cu-cr/sio2 and cu-cr-mo/sio2 catalysts. Among the cu-cr/sio2 catalysts, chromium aids can improve catalyst performance but are less stable; cu-cr-mo/sio2 catalysts have a copper content of between 12 and 18 per cent, catalyst performance and equipment production capacity have increased significantly, production costs have decreased and cu-cr-mo/sio2 catalyst activity is related to cuo。
The effects of preparation methods, component content and reaction process conditions on the reactive performance of nitrobenzene plus hydrogen benzidine on (cu) ceo2 compound catalysts were studied, among others. Nitrobenzene conversion rate and benzidine selectivity reached close to the structural threshold of nearly 100 per cent near the 9 per cent cu mass score in ceo2 (cu) and had good stability. Optimized reaction condition of 190°c, hydrogen pressure 0. 5 mpa, h2/c6h5no2 substance > 4, nitrobenzene feed at an air speed of less than 6h-1。
(2) nickel is a catalyst
The nickel system catalyst, which is mainly used in the hydrogen-composed reaction process of nitrobenzene liquids, including rainey ni catalysts, load-type ni/sio2 catalysts, refined nanonickel and non-crystal alloy nickel catalysts, is low-cost, selective but resistant to poor toxicity, and trace organic sulphides are highly susceptible to catalyst poisoning。
Ni-b/k2ti6o13, which has excellent catalytic and hydrogen-added properties for non-crystalized ni-b catalysts and which have limited the characteristics of their wider application through poor stability, has prepared a load of non-crystal alloy catalysts through impregnated-chemical reduction. Add appropriate k2ti6o13 to non-clinic ni-b catalysts, increasing the activity and stability of non-climate ni-b catalysts. Reactive at 100°c and hydrogen subpressure at 2. 0 mpa at 120min, the conversion rate of nitrobenzene and the selectivity of benzidine can be as high as 99. 5% and 98. 0%, respectively, at the reaction temperature of 100°c, and the conversion rate and selectivity of catalysts after six cycles are 98. 0% and 93. 0%, respectively。
Li gui xian, among others, uses impregnated sedimentary systems for ni/hy catalysts and applies them to the reaction of hydro-synthetic benzo-amphetamine compounds in nitro-based compounds. Ni/hy catalysts have a high catalytic activity, and the rate of transformation of nitrochemicals and the selectivity of benzidine-like compounds is higher than 99. 0 per cent under mild reaction conditions. The catalyst can be stored in an atmosphere below 150°c, with a high dispersion of active components and good magnetic separation。
Wang minghui and others examined the catalytic activity and selectivity of ni-b/sio2 non-clinic catalysts in the reaction to high-voltage liquid nitrobenzene plus hydrogen benzidine. The catalyst is not only highly catalytic, but also highly selective and superior to rainey ni and other ni catalysts. Crystalization leads to the loss of a catalyst, and the presence of a carrier not only increases the dispersion of the catalyst but also stabilizes non-cline structures. Keep the catalyst in ethanol keeps its activity constant。
Von shih hong and others, acting with ultrasound, synthesized the pre-exposures lixmos2 and ni/al-mos2 composites using single-molecular strip-heavy-heavy technology, with an orthobutyl lithium column plug in the mos2 layer. The composite material is used as a catalyst for the hydromethamphetamine reaction in nitrobenzene liquids. Ni/al-mos2 catalysts using n(ni)/n(al)=0. 5 were converted 99. 1 per cent for nitrobenzene and 98. 8 per cent for benzidine under reaction conditions with reaction temperature of 383k, h2 fraction pressure of 2. 0mpa and air speed of 3h-1。
Niso4 6h2o, naoh and h2 were used as raw materials for the preparation of nanonickel powder, which is evenly distributed with an average particle size of 20 to 35 nm, using a low-temperature solid phase. The activity of its catalytic nitrobenzene liquid combined with hydrogen is about nine times greater than that of rainey ni。
(3) precious metal catalyst
Precious metal catalysts include mainly pt, pd and rh. Catalysts of this type have the advantage of high catalytic activity and longevity, but production costs are high. Carrying precious metals are aluminium oxide, activated carbon and high molecules. When precious metal catalysts are used, nitro-biphenyl-catalytic hydrogen is in most cases liquid-catalytic hydrogen。
Liu pu and others, using sio2 carriers, melamine and formaldehyde indents as high molecular formulations, produced a new sio2 high-molecular formulation of nitrogen impregnated impurities, and examined its catalytic hydrogenization of nitrobenzene. In composite carriers, the better nitrogen content is 1. 49 per cent; the catalysts prepared for ethanol reduction under nitrogen protection are highly catalytic. 1 ml nitrobenzene in ethanol solvents at a reaction temperature of 313k and 2h hydrogen under 0. 1mpa pressure under the catalyst of 0. 3g (0. 00523mmol pd/0. 1g catalyst) can convert nitrobenzene to 98%, while the product is only benzidine。




