Many plastics are thermal wood stabilization material. When it is heated in air, degradation reacts often. Release volatile gases, leaving porous residues. Residues are usually composed of carbon sludge, which has the capacity to absorb radiation heat, resulting in cumulative warming. Oxygen in the air is also easily fed into porous residues, and plastics burn when oxygen concentrations, and the temperatures of the residues reach volatile gas combustion temperatures. This places many restrictions on plastic applications。
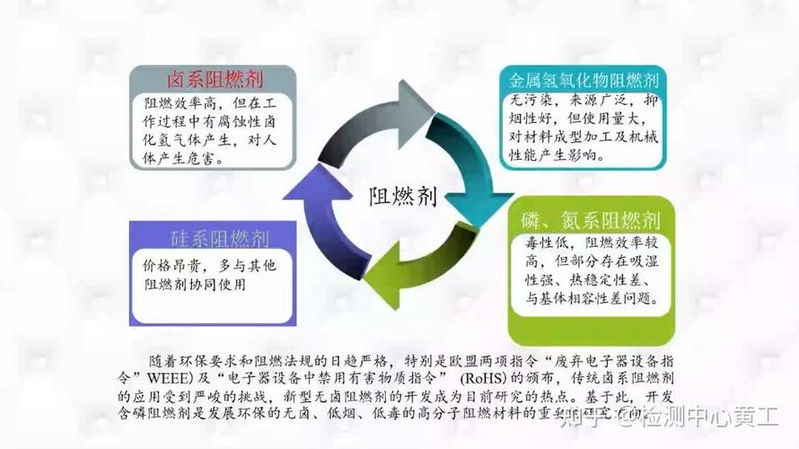
If substances such as phosphorous, halogenated organic matter or antimony trioxide are added to plastics to prevent or slow their combustion, they are referred to as flame retardants. In addition, the introduction of inflammable structural clusters, known as reaction flame retardants, in the synthesis of certain polymers such as epoxy, polyester, polyurethane and abs, can reduce their combustion performance。

Most commonly used flame retardants are compounds of elements such as aluminium, nitrogen, phosphorus, antimony, chlorine, bromine, etc., such as phosphate cheeses such as diphenol phosphate, triphenyl phosphate, diphenyl phosphate, etc., and halogenated phosphate. Category
For example, tri2,3-dibromopropyl phosphate, organic halogenates such as chlorinated paraffins with 70% chlorine, hexabromobenzene, decabromodiphenyl ether, chlorinated biphenyl, inorganic flame retardants such as trioxide, aluminium hydroxide v of magnesium hydroxide, hydrohydrazide, monoponate, zinc borate, crophosphorus, etc。
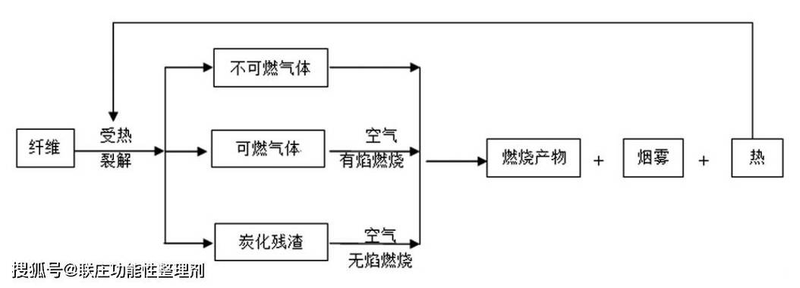
The basic function of flame retardants is to interfere with oxygen, heat and flammables, which are three essential elements for maintaining combustion. This can generally be achieved by the following: 1 flame retardant can produce heavier non-flammable gases or high-boiling point liquids, covered by plastic surfaces, and the link between oxygen and flammables is blocked. 2 reduced surface temperature of polymers by thermal decomposition or heating of flame retardants. Flame retardation produces large quantities of non-flammable gases, which dilutes the flammable concentrations of gases and oxygen concentrations in the burning areas. 4 flame retardants capture active free radicals and interrupt chain oxidation reactions。