Basic definitions commonly used in empty operations
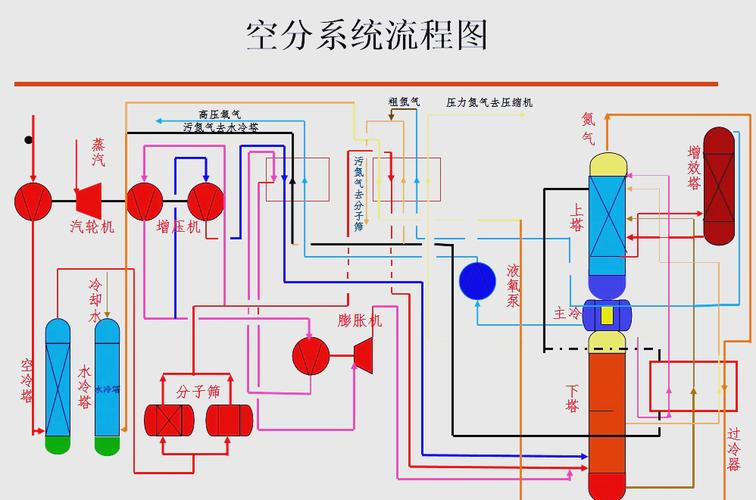
1. Air separation: the process of separating its components from air to produce oxygen, nitrogen and to extract gases such as argon, helium, helium, kryptonium and thorium。
2. Bus: is the rotation of a spare or deactivated rotor through the periodic direction of a person, in order to prevent the rotor of the equipment from deforming in a static state。
3. Gasification subheat: the amount of heat required to transform liquid molecules into vapour molecules at saturated temperatures。
4. Temperature (t): is a sign of the body's coolness, which is related to the body's thermal state of motion。
T(k)=t(°c)+273
K is the international temperature standard, t is the temperature standard.
Optimal gases: gases that strictly comply with the gaseous equation (pv=(m/m)rt=nrt) are called ideal gas (ideal gas. Some books refer to gases that meet the three main laws of the gas.) from a micro-level point of view, are gases that are negligible in size and in vibrancy between the molecules, called ideal gas。
6. State equations for ideal gases:
P1 v1/t1 = p2 v2/t2 =... =pv/t = constant r
Breathing: when centrifugal compressor export pressure rises and flows are reduced to a certain degree, the machine is unstable, the flow is more volatile in a relatively short period of time, and the compressor pressure suddenly falls, it is very volatile, it is highly volatile, it produces a strong vibration and it is called asthma that causes abnormal noise。
8. Heat-replacement equipment: equipment for the transfer of heat from thermal fluids to the cold fluids is called heat-replacement equipment。
9. Optimal power: indicates that no loss is taken into account, that steam is ideal for expansion in a steam engine, and that all heat is converted into power in units。
Depressive adsorption: the process of adsorption using pressure effects is repeated under a higher pressure adsorption and under a lower (or negative) pressure during the adsorption-regeneration operating period。
11. The first law of thermodynamics: all objects in nature are energy, and energy takes different forms, which can be transformed from one form to another, from one object to another, and the sum of energy remains constant during conversion and transfer. It can be turned into power, and it can be turned into heat; it must produce a certain amount of heat, and it must produce a certain amount of power when it disappears。
12. The second law of thermodynamics: the heat cannot be passed from colder objects to more hot objects alone at no cost (without compensation)。
Regeneration: regeneration is the reverse process of adsorption. Since the adsorbent is saturated by the sorbent, it loses the adsorption capacity and measures must be taken to restore the adsorbency of the adsorbent, which is regenerative。
14. Exposure point: the temperature at which the vapour is saturated is called a spot。
15. Saturation content: the concentration of water vapour in air, which is called saturation content, cannot continue to increase。
Adsorption "transformation points": points where the concentration of the sorbent is significantly increased when the fluid is out of the sorbent layer。
17. Throttle: fluids reduce stress by swelling with sharp holes。
18. Salutation effect (jour-tomson effect): temperature changes resulting from inflating the gas。
The effect of entropy: temperature changes due to pressure changes when the gas expands。
20. Upgrade: from solid phase directly to vapour phase transformation。
21. Thermal temperature differential: refers to the temperature difference between cold heat fluids at the heat exchanger end。
Central temperature difference: refers to the temperature difference between cold thermal fluids in the middle of the heat exchanger。
23. Cold end temperature differential: refers to the temperature difference between cold heat fluids at the cold end of the heat exchanger。
Fluid vapour ratio (reflow ratio): the ratio of the volume of current liquids in the distillation tower to the increase in vapour volume。
25. Liquidity: the high rate of increase in vapour in distillation towers prevents normal spilling of liquids。
26. Leakage: in sift-painted distillation towers, leaking liquid from the sift is caused by the low rate of increase in vapour。
27. Run-off cold losses: the cold loss resulting from the temperature difference between equipment working below ambient temperatures and the surrounding medium。
28. Loss of reheating: the temperature differential between heat exchangers and cooling fluids leads to incomplete loss of cold recovery。
Refrigeration losses: this refers to the loss of cooling in the cold of the air separation equipment due to the loss of cold running and the lack of heating。
Evaporation: a carburetorization process at an exposed interface of liquids at a certain temperature。
31. Boiling: there is a carbureting process within the liquid, whereby bubbles within the liquid rise and become steam and run to the upper space of the liquid layer。
Saturation vapour pressure: the number of vapour molecules in space is no longer increasing and the vapour pressure is maintained to a certain degree and balanced。
Saturation temperature: the corresponding liquefied temperature of saturation vapour pressure。
Volatile components: the lower the saturation temperature corresponding to the same pressure, the more easily the substance can be vapourized。
35. Critical temperature: only below this temperature is it possible to liquidate it by increasing pressure, the highest temperature。
Critical pressure: liquefied pressure required at critical temperature。
Overheating vapour: vapour exceeding saturation temperature is also called unsaturated vapour。
Overcooled liquids: liquids whose temperature is lower than that of the pressure and which correspond to the saturated temperature are also called unsaturated liquids。
Fog bands: when the vapour speed is further increased or the plate spacing is too small to have enough space to separate the gas from the fluid, the steam packs the liquids up to another tower plate, thus reducing the efficiency of the tower plate, which is described as the fog bands, which are not allowed in the sift tower design。
Phase: if the parameter values of the system state are consistent at all points of the system or change continuously without mutations, the system is called a single-phase or one-size-fits-all system. Substances of a certain quantity and of a physical nature in the whole mass are referred to as objects. All parts are in the same state if they are divided into parts by any method。
41. Balancing of gaseous fluids: in closed packagings, under certain conditions, some molecules in the various components of the liquid phase escape from the interface into the gas phase space above the surface of the liquid, while some molecules in the gas phase return to the liquid phase. After prolonged exposure, the process is balanced when the molecules of each group escape from the liquid phase at the same speed as the gas phase, or when the balance is achieved。
42. Oxygen extraction rate: ratio of total oxygen in product oxygen to total oxygen in process air in feed towers
焓: 焓 is the sum of inner and mobile energy in the fluid, expressed as symbol h
44. Entropy: entropy is the condition parameter of the gas, with a certain entropy value when the pressure of the gas remains constant. Expressed as sign s. When the gas gets heat, the entropy increases, and the increased entropy s equals the temperature of the calcium q divided by the temperature of the gas, t, i. E. Δq/t. In short, entropy is a measure of price differentials in both states。
45. Distillation: the principle of distillation is to evaporate parts of the condensed and mixed liquids over time, using different boiling points for both substances。
46. Cooling: cooling usually refers to the absorption capacity of cryogenic objects relative to the surrounding environment。




