You know, salt uses a lot of things, and besides being edible, it's one of the main catalysts for artificial rainfall。
So, do you think you can come up with the idea of catching a salt in the kitchen and spilling it over the clouds in the air for the purpose of increasing the rain
Actually, no. Generally, the diameter of an edible salt grain is approximately 0. 1 mm, or about 100 micrometres. This is a very small part of the food. But for air vapours, a salt-eating particle is a huge guy because, in clouds that do not produce rainfall, generally the size of the water vapour molecule is only 5-10 μm. A grain of salt is thrown into the clouds in the air, like a ball of lead thrown on marshmallows, which, due to gravity, has reached the drop standard and goes straight through the cloud. So it can be assumed that when the operator throws a piece of salt into the cloud, the salt will fall and the cloud will fall。
So how does salt make it rain on clouds
Salt is used mainly for warm clouds and rain. When warm clouds naturally fall, clouds drop more than 100 μm in diameter in order to defeat the rising current and form rain drops to the ground. When water vapours are abundant in warm clouds, water vapour particles typically have a diameter of between 5 and 10 micrometres, which are freely suspended in the air because they are very light and do not fall down. If the time is taken to sow salt powder into the clouds, because the arrival of salt powder unites water vapour particles, and when the diameter of the water vapour particles increases to dozens of micrometres, they continue to collide with the surrounding small droplets, eventually overcoming the rising current and becoming rain drops。
It is therefore true that salt is used as a humid agent to increase the rain, and it is old. Just to increase the rain, you have to do "skinned" work on salt particles. Salt grains that add rain to the warm clouds at an early stage are grinded by physical methods until their diameter is about 30 um. But because of the size of the head, when the salt powder is thrown into the clouds, the effect of the rain is not very good。
After repeated practice, the chemical method is now used to produce a salt particle of less than 1 μm (pfc, sodium chloride) by means of combustion between substances. Because of their small size, these salt particles are suitable for play with water vapour in the clouds, and they are sent into the clouds, so that they can be closely associated with the water vapour molecules, and in the process they continue to wet, collide and grow. Experimental evidence has shown that the more salt powder is soaked, the smaller the diameter of the salt, the more rain is produced。
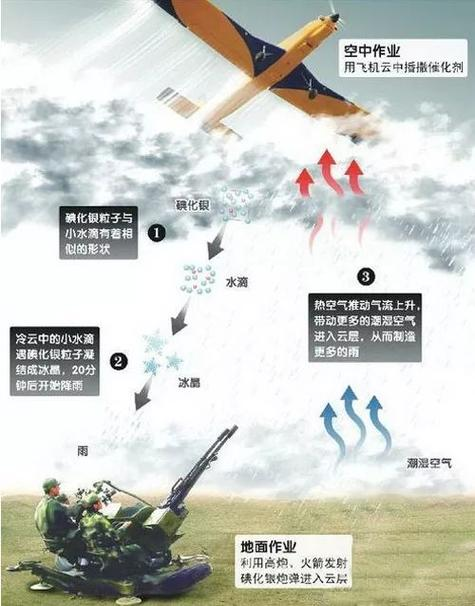
In practice, it is more common for cold clouds to be rained by manual methods. Generally speaking, through catalysts, the ice crystals in the clouds are increasing and increasing, and eventually they melt into rain. The catalysts for cold clouds and rain are icy agents (e. G. Silver iodide) and refrigerants (e. G. Dry ice, liquid carbon dioxide, liquid nitrogen)。

In conclusion, artificially influenced weather catalysts are classified into ice agents, refrigerants and damps。
Ice: cold cloud catalytic operations, mainly for 6°c ~-20°c. Silver iodide is one of the most commonly used ice agents with a crystal structure similar to that of ice crystals and a master in making ice crystals. Silver iodization microparticles are obtained mainly by chemical means, and on human-image aircraft, staff light a percentage of the chemical composition and accelerants. When temperatures above 2000°c are reached, the resulting silver iodization gassed in an instant, cold and fast condensed air, and small silver iodized particles immediately enter the cloud into working conditions and become ice. In general, 1013 to 1014 silver iodized particles can be separated per gram of silver iodized。
Refrigerant: for fluorinated cloud or layered cloud catalytic operations at 0°c ~-6°c with members such as dry ice, liquid nitrogen, propane, etc. Dry ice is solid carbon dioxide, with an elevated temperature of -78. 5°c, which increases directly from solid to gaseous, and a g dry ice that produces tens of billions of crystals, a cooler. The boiling point for liquid nitrogen is 195. 8°c and the boiling point for propane is -80°c, working in a similar manner to dry ice。
Wetizer: for heating clouds above 0°c. With members such as sodium chloride (salt), calcium chloride, ammonium nitrate and urea, one g of salt can catalyse tens of millions of raindrop embryos。

In terms of weight, a 1 gram catalyst can trigger 100 to tens of billions of ice cores or raindrop embryos, crystals, so that only a small amount of catalyst is required for each operation to perform the task efficiently。
In terms of components, such as coolant dry ice, liquid nitrogen, gasification becomes carbon dioxide and nitrogen gas - all of which form part of the air - after participating in the rain surge. Then, for example, weeding salt, urea, and so on, they originate in nature and return to nature after the rain. So they are both ecologically safe catalysts and certainly do not pollute the environment。
For example, the use of more frequent silver iodization, the use of both rain-added and rocket operations by aircraft is very small, and long-term data monitoring shows that the accumulation of silver ion concentrations in water and soil in the area of operations is much lower than those specified by the world health organization。
Therefore, the correct use of human-image catalysts does not cause environmental pollution。




