The main principle of electrolytic hydrogen is that water molecules are released from direct current power to produce oxygen and hydrogen gas, which are analysed from the anode and cathodes of the electrolytic tank, respectively. Hydrogen electrolyte is an efficient and clean hydrogen production technology with simple hydrogen production processes, high product purity and generally 99. 9 per cent hydrogen and oxygen purity, among the most promising large-scale hydrogen production technologies. The hydrolysis response can be divided into two semi-reactions of oer and her, with the following specific mechanisms:
01
Oer mechanism
Hydrogen, as a high-energy-density zero carbon energy carrier, is considered a promising sustainable energy source. There is widespread interest in electrolyte hydrolysis of hydrogen. Among them, oer involves a multi-electronic transfer process, and the slowness of the kinetic response is a key constraint on the efficiency of the entire hydrolysis unit. By studying its response mechanisms, more efficient catalyst materials can be designed in theory. The oer mechanism involves adsorption and desorption of intermediates, i. E. Ohads→ooohads→o2ads process (aem mechanism for adsorption product evolution). In alkaline and acidic media, the oer mechanism consists of four steps (formula 1 to 12) (each of which is co-ordinated with an electronic) and involves multiple intermediates (ohads, oads, oohads and o2ads). In alkaline and acidic media, the liberty of gibbs required for oer reaction is 4. 92 ev (type 6 or 12) and the freedom of gibbs required for two intermediate steps is 3. 2 ev (forms 2 and 3 or 9 and 10). If one of these two steps requires a lower guibus free capacity, then it should be a higher gibbs free process in the other. The follow-up to the high level of freedom and power in gibbs is the decision on the oer rate (rds), and vice versa。
Under alkaline conditions oer's base dollar response steps are as follows:
Oh-+ *ohads + e-(1)
Ohads + oh-oads + h2o + e-(2)
Oads+oh-oohads+e-(3)
Oohads + oh-o2ads + h2o + e-(4)
O2ads→o2+*(5)
Total reaction: 4ah-→ 2h2o + o2 + 4e-(6)
Under the acidic conditions, oer's base dollar response steps are as follows:
H2o + *ohads + h+ e-(7)
Ohads + oads + h+ e-8
Oads + h2o →oohads + h+ e- (9)
Oohads + o2adds + h+ e- (10)
O2ads→o2+* (11)
Total reaction: 2h2o → 4h+ + o2 + 4e-(12)
It* indicates the active position of the surface of the catalyst, and “ads” indicates the adsorption status of intermediate products (ohads, oohads, o2ads)。
Typically, m-o-key interactions have a significant effect on the stability of the catalyst's surface middle and have a significant impact on the overall electro-catalytic efficiency during the catalytic oer process of the metal (m) catalyst (including precious and transitional metals). In alkaline and acidic media, oer response mechanisms can be written as follows (see figure 1 for further details) [1]. Blue lines represent acid/orange lines and oer response mechanisms under alkaline conditions. The black line indicates that o's evolution involves the formation of an intermediate hydrogen peroxide (m-oah) and that two adjacent oxygen (m-o) directly reacts to the production of oxygen (green) (intermediate green arrow indicates the likelihood of producing oxygen, not the m-ooh intermediate)。
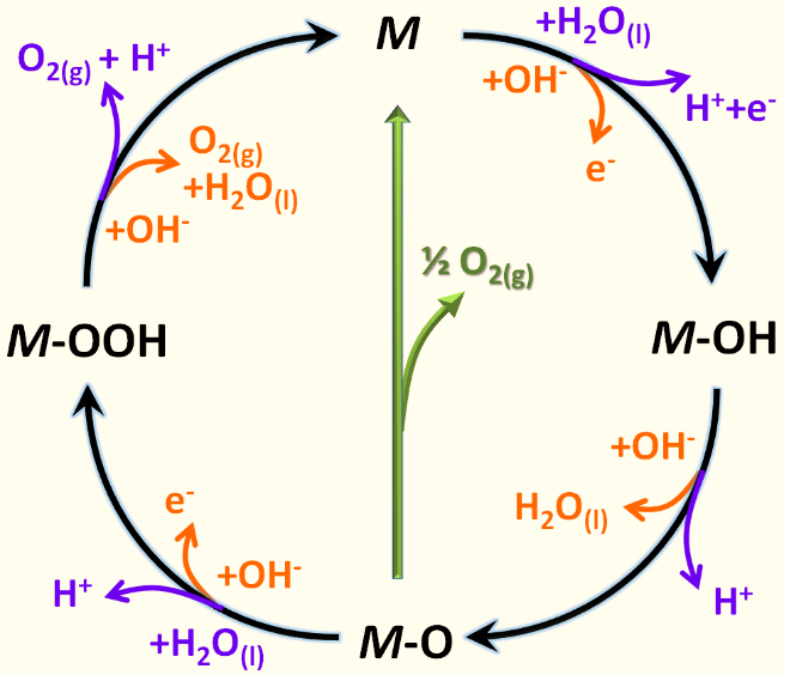
Figure 1。

Under standard conditions, oer has a thermodynamic balance of 1. 23 v, but there are adverse dynamics that hinder reactions in the actual reaction, and an additional force is required, namely, electricity overpower. As noted above, these four steps in thermodynamics are non-autonomous and require considerable energy to move forward. The power force is closely related to each step of oer, which prevents kinetics and requires a higher power level to overcome the energy force. The resolution of the energy relationship between ohads and oohads was seen as key to achieving the desired conditions for low power levels。
02
Her mechanism
The catalytic activity of the hydrogen reaction (her) is related to the adsorption of the hydrogen gas (Δgh*). Her can be based on the volmer-heyrovsky or volmer-tafel mechanisms (as shown in figure 2) [1]. In the alkaline (figure 2a) or acidic (figure 2b) media, the evolutionary steps of her are similar. The first step of the her is to react with h3o+ in acidic media to produce h* and with h2o in alkaline media to generate h* (see formulas 13 and 16). The second step is the rds, which depends on the activity of these catalysts (see formula 14, 15, 17, 18). For example, pt-based catalysts, tafel reacts to adsorption h* and h* to generate h2, while transition metal catalysts typically generate h2 with h2o (alkaline) or h+, i. E. H3o+ (acidity) and h*. The reason for the different response paths is that the gill gh* on the pt metal is close to zero, while the gill gh* on the transition metal is very high。
Under alkaline conditions, the ger's base dollar response steps are as follows:
H2o + e- → oh-+ hads (volmer) (13)
Hads + h2o + e- → oh-+h2 (heyrovsky) (14)
Or 2hads → h2 (tafel) (15)
Under the acidic conditions, the basic meta-response steps for her are as follows:
H++e-+* hads (volmer) (16)
Hads + h+ e-h2 (heyrovsky) (17)
Or 2hads → h2 (tafel) (18)
It* indicates the active biting of the surface of the catalyst and the ads indicates the adsorption of the intermediate (hads)。
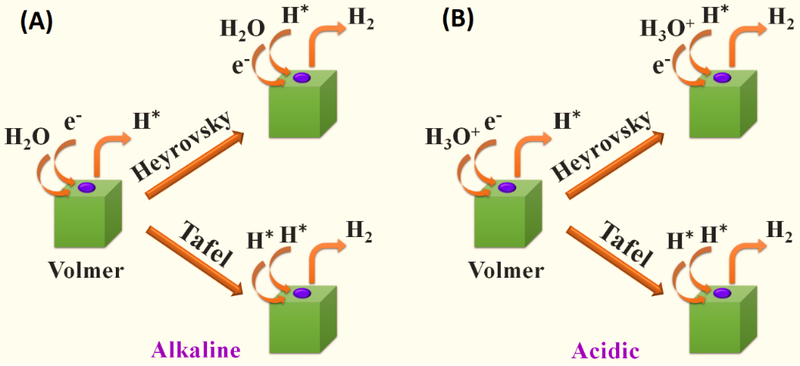
Figure 2。
Hydrogen adsorption capacity is one of the important factors to be taken into account when designing hydrogen-energy catalysts. For example, the single layer of ptsex non-crystal catalyst displays excellent her catalytic performance (0 v throughput, 39 mv dec-1) similar to the pure pt surface catalyst (0 v through solid phase, 37 mv dec-1) (see figure 3) [2]. The ptsex catalyst has a high-density uniatomic layer structure (figure 3a), showing a high tafel reaction mechanism (chemical step) in step 2 (figure 3b). In particular, theoretical calculations suggest that non-clinical super thin structures can significantly improve the hydrogen adsorption performance of catalysts compared to crystal structure catalysts. Of these, the monolayers of non-crystal ptsex catalysts are located near 0 ev, and the original crystal ptse2 catalysts are above 1 ev. Other non-crystal catalysts (e. G., non-crystal pd quantum points) showed similar enhancements in her (tafel tilt of 30. 0 mv dec-1)。
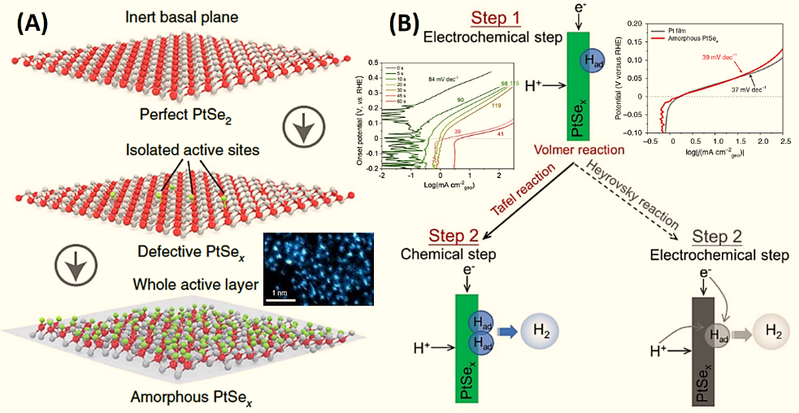
Figure 3。
03