Start with the organic chemical reaction machine
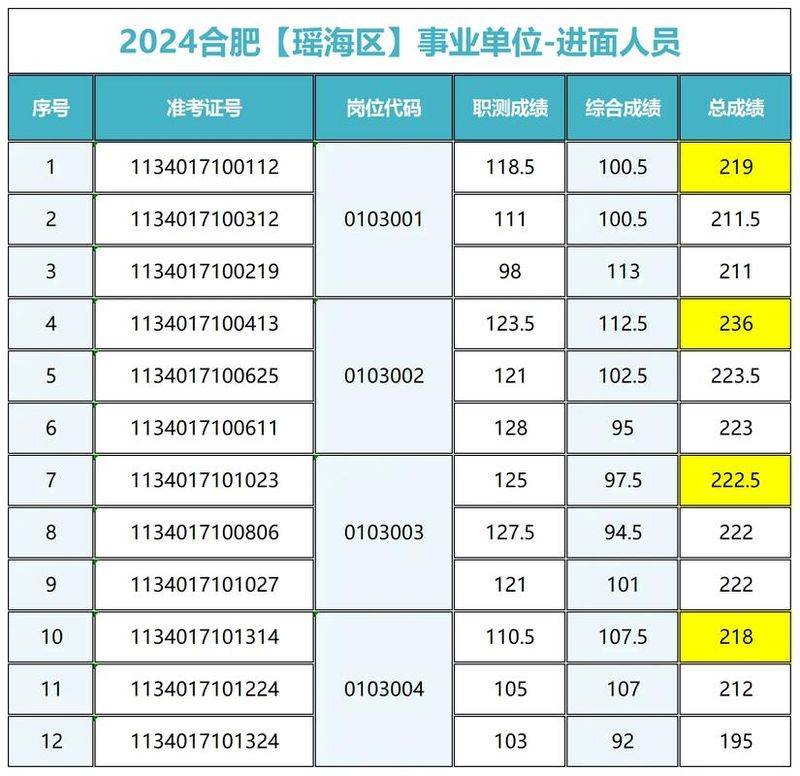
It really felt like it was a mess, with a few arrows pointing at it and not knowing what it meant, listening to it and not learning it. After an organic study, i can't figure out how to do it, i'm happy to get up, i can't work and i'm slowly paying back my debts to school。
Reaction mechanisms can be understood as processes in which chemical reactions occur, including molecular fractures and formations, electronic transfers, and intermediates and transition-states involved in the process, a continuous description of the reaction to the product。
Some of the concepts involved:
Basic reactions: these are inseparable, the simplest response steps. A complex response usually consists of many meta-responses。
Intermediates: substances that are temporarily formed during the reaction and then consumed in subsequent reactions。
Transition states: unstable state of instantaneous formation and consumption during reaction. It often plays a decisive role in response rates。
The most slow step in the response determines the rate of the overall response。
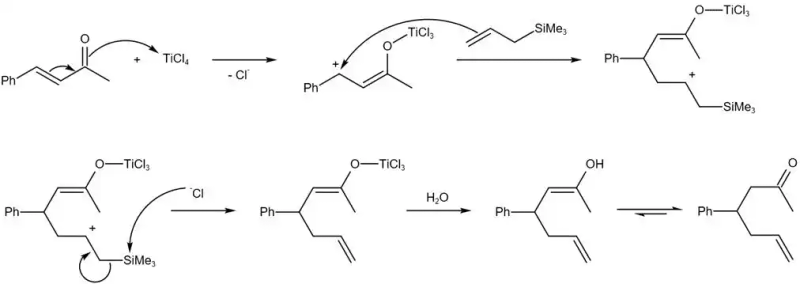
What about the reactor
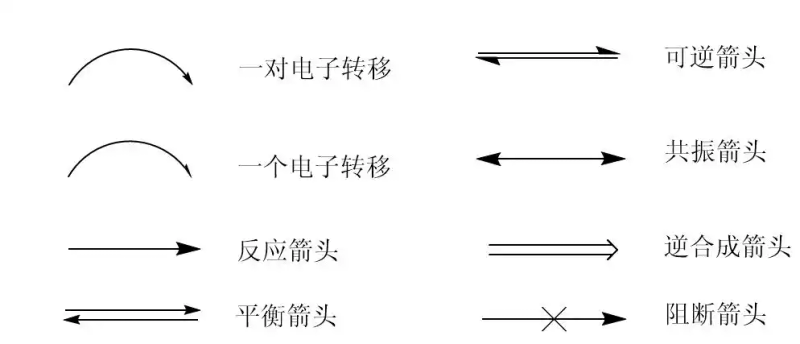
What's an arrow
Let's take a look at some of the arrows we used to use
Bend arrows: a shift in electronics
Fish hook arrows: transfer on behalf of an electron
Reaction arrow: reaction
Balance arrow: response balance
Reversible arrows: reversible
Resonance cutting: resonance, mostly in co-conforming structural analysis
Counter-synthetic arrows: reverse synthesis, product to reaction
Block arrows: no reaction
What's the negative
The negative is a form charge, which by definition can really be understood as a form. Nevertheless, it still plays an extremely important role in chemistry, especially in organic chemistry。
At the macro level, the introduction of form charges makes lewis'structure more precise and rational, making it one of the key components of the structure of louis; at the micro level, form charges are agreed to in accordance with the octet rule。
Therefore, form charge is not an accurate charge state (e. G. Positive or negative charge) or real charge distribution position, which is significantly different from partial charge center (partial charge center)。
While form charge is not an absolute indicator of the actual charge state, it is not without practical effect。
The presence of form charges has facilitated chemical structure analysis and reactor extrapolation, effectively simplifying these processes. Analysis of form charge allows us to predict the distribution of electrons in molecules, thus understanding the likelihood and direction of reactions and providing important clues for research on chemical reactions。
One more chestnut
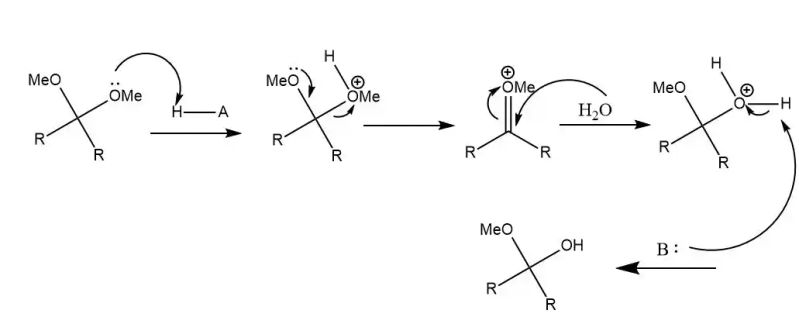
For example, in one reaction, the oxygen ion of the electron offensive acid received the second structure; then another oxytocin transferred the oxytocin to the carbon dioxide, forming the cylindrical key, allowing the oxytocin to be transferred to the oxygen atom, the third structure; in water molecules, the oxytocin atoms moved to the cytoac, the oxytocin to the oxygen atom, and the cytocin to the oxygen atoms received the structure four; the alkaline attacked the hydrogen atom again, the hydrogen atom departed, the hydrogen atoms moved to the oxygen, the hydrogen atoms moved to the oxygen, and obtained the products, the structure five。
How do you draw a reaction machine
Identification of reactions and generators
At first glance, that's bullshit, but what we're talking about here is a reaction and a product of a step-by-step response. This means disaggregating the reaction as far as possible and identifying the intermediates involved in the reaction, transitioning the state and establishing a complete response path。
There's a paradox here, and if i could analyze all the reactions and figure out what the mechanism is, i'd have done it because i didn't understand it, but you told me that the first step of the mechanism was to dismantle it. In fact, there is no need to be too worried about the mechanisms of most organic chemical reactions, which require a period of familiarization and accumulation and cannot be stopped。
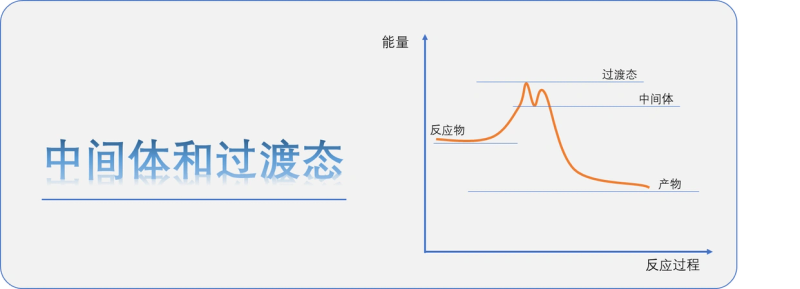
Insert a knowledge point, distinction between intermediate and transitional states:
The intermediate is a relatively stable substance that can be observed at least to some extent. In the course of its response, the reactor is transformed into an intermediate state of the product, which may exist as a stand-alone substance, with a certain configuration, which may be further reacted to or may even be separated from other substances. Moreover, intermediates can exist for a much longer period of time than transition states, and concentrations can be observed and can be preserved and transferred in reactors。
The transitional state is a state of instantaneous reaction between the products, the highest energy point of the reaction system and the most unstable moment of reaction. It cannot be directly observed and is generally obtained only by calculating chemistry. At this point, the chemical reaction system is dynamically balanced and there are no stable chemical keys between the reaction and the product. It exists for an extremely short period of time and cannot generally be separated。
Confirm homo and lumo
The front-line orbit theory (explanation of homo and lomo) can be consulted in my previous response
In fact, the first electronic transfer arrow, which represents the re-orbiting process, homo→lumo, requires an analysis of the entire reaction to begin drawing the whole mechanism, which, in the words of the word, is to find the starting point, and to put the baby back together one by one。
Push electrons
When the electron is pushed through an arrow, it is discovered that some atoms or chemical keys are more electronic than they can fit and that they do not conform to the eight cylindrical rules. So to whom the electron is pushed, of course it's close enough to load the electron atom. How many electrons do we push? You can try to analyze the top example。
Common errors and notes
Electronic shell overload
It's still easier to show, and the electrons push it around and push it. Bearing in mind cycle two, 8 electronic, and cycle three, 18 electronic。
There's a molecular reaction
A picture, intuitively, that says that the three molecules have a very low probability of collision and reaction at the same time, is an unreasonable description。
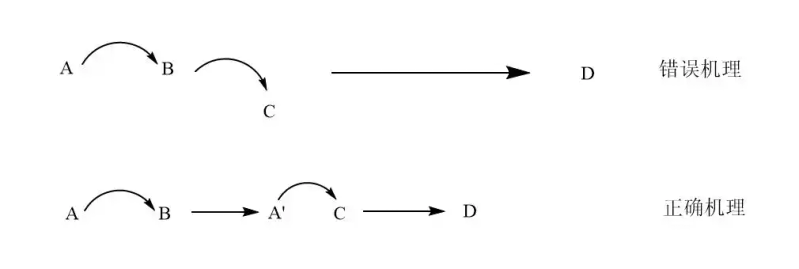
Arrows are greedy
That is, the reaction split is not in place, it is not decomposition into a base dollar response, and it is generally reasonable to move up to three arrows。
Conditional error
For example, under alkaline conditions, ha, under acidic conditions, b: in solution, h+ means hydrogen ion (should be h3o+, or directly ha)。
What's the use of learning how to do that
Projected response results:
Reaction mechanisms can help us predict chemical reactions. By looking at how the chemical key breaks and how the new compound is formed, we can predict the product of the reaction and the probability of its generation, the primary by-product。
Optimizing the response process:
Knowledge of the response mechanisms optimizes the response process, such as increasing the rate of response and increasing product selectivity. If, for example, we know that a certain step is the rate of response that determines the step, then we can explore different conditions or use catalysts to accelerate it。
Designing new responses and structures:
Response mechanisms are critical to the development of new structures to construct new responses, which may be used to prepare new chemicals or to analyse existing structures. In particular, in drug development, more effective drugs can be designed by understanding the coping mechanisms of drug molecules and biological targets。
Let's hope it helps. Let's move forward. Come on