Lithium ion cells are subject to a certain expansion and contraction during the recharge cycle, and this repeated volume change can result in the destruction of the polar tablets by powdering, stripping, etc., thus affecting battery performance. Therefore, improving and maintaining negative viscosity contributes to achieving stable battery performance。
In order to optimize the negative viscosity, the study of negative viscosity factors based on the negative viscosity of polyvinyl fluoride (pvdf) viscosity is conducted in this paper, mainly in relation to the viscosity of copper platitude and surface state, environmental moisture, electrolytic fluid moisture, etc。
Experiment
1. 1 materials and polar film preparation
Materials: commercial electrolyte 1 mo/l lipf6/(ec+dmc+emc) (mass equal to 1⁄1), man-made graphite, conductor sub-p, pvdf, nmp, etc., are battery-grade, electrolyte copper plating, and condensed copper plating。
(b) polar preparation: negative polar slurry made of w (pvdf) ∶w (conductor) ∶w (graphite) = 3. 5 ∶2 > 94. 5 and soluble in nmp (acrylic acid additive), coated in copper slurry, then baked and crushed into negative polar slices。
1. 2 instruments and tests
The copper scanned mirror (sem) was used for surface profiling tests. Tests on the distribution of copper aluminum surface elements using the energy dispersion spectra (eds). A multi-functional electronic puller is used to conduct a viscosity test with a polar width of 4. 5 cm and a length of 15 cm。
Results and discussions
2. 1 distinction between electrolyte and condensed copper plating
Figure 1 (a) shows the microform image of the inside of the rolls of copper; figure 1 (b) shows the microforms outside the rolls of coppers, and the squeezing stripes of the squeezing of coppers are more visible when they are crushed, and there is no significant difference between the sides of the squeezings. Figure 1 (c) shows the microforms of the electrolytic copper plating face, while figure 1 (d) shows the microforms of the plating plating face, with a marked difference between the plating face of the plating face of the plating face, the surface of the plating face being the outer surface of the plating surface, the plating plating face matching that of the plating face, and the plating pluming of the plume surface being the relatively smooth surface of the plume of the plume by a plume-grade crystal stack。

There are not only significant differences in microforms between the pressured and electrolytic copper beams, but also differences in the surface chemistry of the copper beams due to differences in the two copper beryllium preparation processes。
In order to compare the differences in the surface state of copper beryllium and electrolyte copper beryllium, the two copper berylliums are treated with herbic acid. At the same time, the treatment of herbic acid allows the active improvement of the surface of the copper aluminum and the erosion of the surface of the copper aluminum, and increases the oxygen content of the corrosive aerobic composition. Bronze is polarized, one is adsorbed to the copper surface, and the other is adsorbed to the powder composition, improving viscosity and thus the desalinization。
Figure 2 shows the state of the surface following the treatment of copper plating and electrolysed copper plating acid. Figure 2 shows the state of the surface of the plating copper plating, c and e, respectively, the plating plating of copper plating, the plating of electrolytic plume, the plume of copper pluming, the plume of electrolytic plume, and the surface of the pyrolytification plating and drying, respectively. As shown in figure 2, the surfaces of the impregnated copper plumes are reded after they are immersed and dried with herbal acid, while the surfaces of the electrolytic plume, electrolytic plume are immersed and dry with herbs. It is known from the results of the above-mentioned experiments that the condition of the pressured copper beryllium is easier to improve, while the surface of the electrolyte copper beryllium is more stable due to the special processes of the electrolytic process and the surface treatment process and is less susceptible to improved treatment with herbal acid。

2. 2 optical viscosity of electrolytic copper plating in comparison to the condensed copper plating
To compare the effect of electrolyte copper plating on the negative viscosity of the pvdf base, polar tablet preparation and viscosity tests are performed. Figure 3 shows the results of the experiments on electrolytic photo-photography and pressure-spray stripping of the inner plaster. In figure 3 (a), the crushing density of the polar tablets is 1. 7 g/cm3, a, b, c and d, respectively, do not dry the electrolytic copper face, dry at 90°c, dry at 110°c, dry at 130°c and strip off the surface of the copper platinum powder。
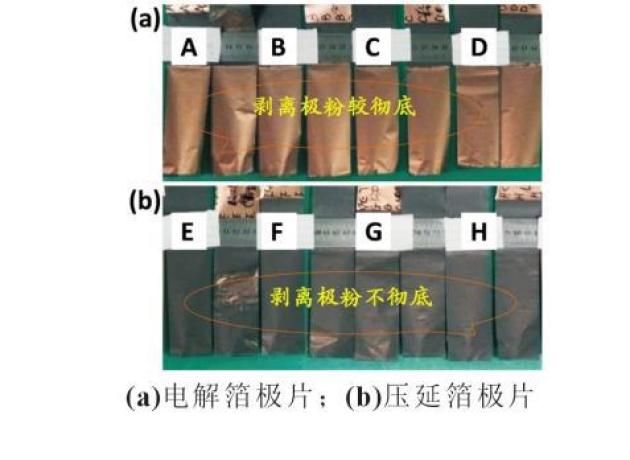
Figure 3 (b), e, f, g and h, respectively, do not bake the inside of the copper bar, bake at 90°c, bake at 110°c, and strip the surface of the copper bar after 130°c. As can be seen from figure 3, the platinum powder is stripped off during visceral viscosity tests, the platinum face of the platinum is removed from the platinum and there are no visible platinum residues; the platinum platinum powder is pressured off the platinum and there are plasters on the platinum. The results show that, under equivalent process conditions, the viscosity of the pressure of copper and powder is stronger than the electrolyte。
Based on the above results, the reasons for the difference between the electrolytic copper plating and the polar glue of the plating copper plating were speculated. First, the surface appearance differences of the two copper beams mentioned above may result in differences in viscosity: following the crushing of the copper plating, the ditch is more visible, and the adhesive flows into the hole on the surface of the copper plume, through molecular proliferation, creeping, etc., to the effect of anchoring. Second, differences in surface status due to differences in copper preparation processes are likely to be the main cause of bonding differences: the viscosity of the pvdf is mainly achieved through inter-molecular force, while the treatment of herbic acid is followed by pressure of the copper aluminum, which enriches the base of the copper aluminum surface and creates a stronger effect with the pvdf; the resistance of the electrolytic copper aluminum is stronger than that of the oxidation of the copper aluminum, and therefore the surface is weaker than that of the copper aluminum。
2. 3 effects of leaching expansion on polar glue
The electrolyte will be absorbed in the polar tablets after the battery is injected, and its viscosity will be affected. To examine the effects of impregnated electrolyte on negative adhesiveness, a polar leachate experiment was conducted. Figure 4 shows a map of the surface of the copper platinum after the disassembly of the electrolytic copper platinum and the pyrolytic pyrolytic platinum. Figure 4 (a) shows, respectively, the non-drying of electrolytic copper aces by a, b, c and d, and dry by 90 °c, 110 °c dry by 130 °c dry by impregnated electrolyte and wind dry by stripping off the surface of the copper acetate; e, f, g and h, respectively, barfing of the inner aces of the copper aces by no baking, 90 °c baked by 90 °c, 110 °c baked by baked by 130 °c, leaching of the electrolyte by drying by wind, and stripping off the surface of the copper acetex。

Figure 4 (b) shows i, j, k and l, respectively, for electrolytic copper plume, pressured copper plume, photolytic plume for electrolytic copper plume, unpressed impregnated impregnated electrolyte outside the plume, and wind drying; and m, n, o and p, respectively, for electrolytic copper plume, pressured for internal plume, electrolytic plume for electrolytic plume, pressured plume for impregnated plume plume, and dry for impregnated plume plating. As can be seen from figure 4, before and after crushing, the electrolytic copper plume/photo skin is more thorough and there are no visible platinum residues; the impurity of the platinum is prolonged / the impurity of the platinum is incomplete and there are pep residues in the platinum. The results confirmed that, under equivalent process conditions, the adhesive force of the condensed copper and powder was stronger than the electrolyte。
The results of the polar viscosity test showed that, following impregnated electrolytic fluids, the viscosity of the electrolytes was reduced to 0. 35 to 0. 50 n before the crushing of the face of copper and the viscosity of the plate before the crushing of the face, to 0. 15 to 0. 35 n after the crushing of the pole; the viscosity of the two-sided crushing of the copper beam was reduced to 0. 40 to 0. 60 n after the crushing of the plate, to approximately 0. 25 to 0. 40 n. The results of the viscosity test of the polar crushing experiment show that the polar impregnated electrolyte causes a decrease in the viscosity of the polar tablets, presumably because the small molecules of solvents in the electrolyte are impregnated into the high molecules (pvdfs), resulting in the melting of the high molecules, the entanglements, the chain movement, the dissolution of the pvdf, and the movement of the entire molecular chain, leading to a decrease in the viscosity of the pvdf。
2. 4 effects of moisture on viscosity
In addition to the copper plating surface/surface state, polar composition, polar tablet preparation process and electrolytic fluids, moisture is an important influence of the viscosity of the polar sheet. The effects of environmental moisture and electrolytic fluid moisture on polar viscosity were studied using the electrolyte copper graphite graphite negative polar system。
Figure 5 shows the shape of the surface around 1h in a 50 per cent humidity environment of negative copper plating at different crushing density. Figures 5(a) to 5(c) are photographs of an electrolyte copper balconium face negatives of 1. 6, 1. 7, 1. 8g/cm3, respectively, with a crush density of 1h before 50% humidity, and 5(d) to 5(f) photographs of an electrolyte radium negatives of 1. 6, 1. 7, 1. 8g/cm 3, with a crush density of 1h after 50% humidity。

As figure 5 shows, after placing 1h in 50% humidity, there is a very pink skin rise in 1. 6, 1. 7, 1. 8 g/cm3 and the very high and low pressure density of the extremes is not the main reason for the very pink skin rise. The results of the experiment show that high environmental moisture content results in the reduction of polar skins, i. E., the viscosity of the polar。
In order to confirm the correlation between pvdf, electrolytic fluids, moisture and polar stripping, blank experiments were designed, with dripping and immersion experiments on fresh negative polar areas of unimpregnated electrolytes, as shown in figure 6, showing no drums of direct dripping or impregnated water from polar unimpregnated electrolysed electrolytes and that the polar areas were hydrophobic (a portion of the polar area remained dry)。

The results of the above-mentioned experiments show that: the polar impregnated electrolyte does not fall powder without moisture; the polar is not immersed in electrolytic fluids, is directly immersed in water and the polar is not powdered; and the polar is dropped by droplets or by impregnation. Thus, the reason for the extreme powdering can be attributed to the fact that after the polar immersion, the pvdf glue is hydrophobic, and when a sufficient amount of water is present, the pvdf after the hydrosis is separated from each other, leading to a dichotomy reaction, which in turn reduces the viscosity of the glue on the copper platinum, so that the environmental moisture can cause the pvdf base to become less adhesive and lead to the extreme powder to be removed from the copper plume。
The pvdf phase separation mechanism was studied, i. E. The pvdf was dissolved in small molecular organic solvents and the pvdf was separated in water after the pvdf was dehydrated. Figure 7 (a) is the phase separation after the pvdf@nmp soluble encounter, and the pvdf condensate from nmp/water solution after the pvdf@nmp solution encounters water, and figure 7 (b)-7 (c) is a schematic for the pvdf solubility mechanism, with small solvent molecules able to penetrate into high molecules, inflate the high molecule, entanglement, chain movement, pvdf solubility, prompting the entire molecular chain movement, leading to a decrease in the binding power of the pvdf。
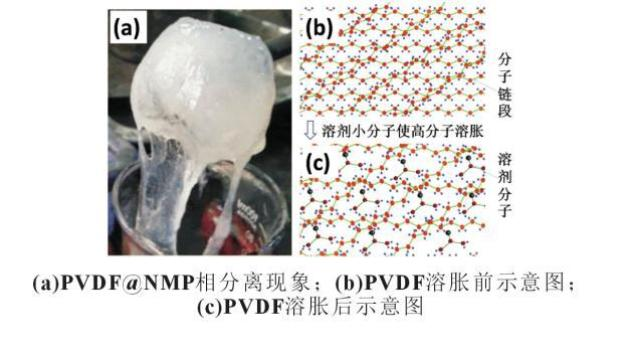
In combination with previous experimental phenomena, dry polar bubble water was not powdered, and impregnated electrolytic fluids were exposed to water skins, so the prosthesis causes: electrolytic micro-molecular molecules cause pvdf to expand/dissolve, dissolved/dissolved pvdfs to a high degree of hydrophobicity, and water separations in the environment lead to the dichotomy of copper。
In addition to the environmental moisture, the effect of the moisture content in the electrolyte on the viscosity of the polar tablet was examined. First, electrolytic fluids with different water content are prepared, then the polar tablets are immersed in different hydrolytic fluids for seven days and changes in the surface state of the polar tablets in different hydroelectric fluids are examined. Figure 8 shows the polar state of the hydrolyte。

Figure 1 (a) is the electrolyte leachate experiment for water content of 34x10-6; figure 1 (b) is the water content of 70x10-6 electrolyte leachate experiment; figure 1 (c) is the water content of 140x10-6 electrolyte leachate experiment; and figure 1 (d) is the water content of 250x10-6 electrolyte leachate experiment. The results of the experiment show that, in figure 1 (a) — figure 1 (c), seven days after the polar immersion, no rise was observed in the polar table, while in figure 1 (d), when water levels reached 250 x 10-6, the polar surface found a slight rise. Experimental results indicate that electrolyte moisture can result in poor viscosity of the pvdf base's negative viscosity and that lithium ion cell electrolytic fluids need to be controlled at lower levels。
Conclusions
The main reason for the difference between the viscosity of the electrolytic copper plating and the pressure-relayed copper plating is the surface form of the copper plume and the extent to which it is coarsed. In addition, environmental moisture and electrolytic fluid moisture can lead to a decrease in polar viscosity. The melting of the polar leachate, the hydrophobic effect of the pvdf glue, which causes a dichotomy reaction, reduces the adhesive force of the glue on the copper platinum, and the environmental moisture and electrolytic fluid moisture can result in the deterioration of the pvdf base negative viscosity. The results of this paper will provide guidance on the process of preparation of polar tablets, hydrolysis control of electrolytic fluids and environmental moisture control when the battery is injected or opened。