Basic requirements
1. The chinese medicine tablets covered by the present technical guidelines shall be of a variety of types with clinical needs of chinese medicine and a history of use in the region。
2. Medicines for standard research on chinese tablets shall be collected in accordance with the standard prescribed for medicines。
3. Except as otherwise provided, the chinese medicines standard institute applies the provisions of the chinese pharmacy by using terms, symbols, units of measurement, general rules codes, testing methods and related requirements。
Standard substances for research purposes shall be certified standard substances issued by the chinese institute of food and drug inspection and other sources。
5. All original records, photographs, etc. Should be kept on file and accessible during the study。
Technical research requirements
(i) title and basis
The title should be based on sufficient justification to clarify the course and the need for revision of the drinking tablet standard。
(ii) artillery process research
1. Original pharmacological requirements
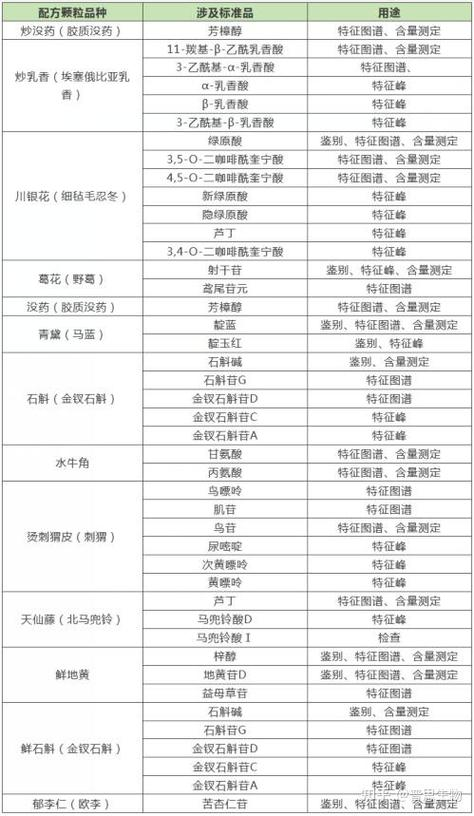
The quality of chinese medicine used in standard studies on drinking tablets should be representative。
2. Artillery auxiliary requirements
Artillery supplements should be accompanied by criteria and sources。
Technical requirements for the artillery process
Artillery methods and key process parameters (e. G. Temperature, time, ancillary usage, etc.) should be studied to justify their reasonableness。
(iii) quality standards studies
The drinking tablet quality criteria include, inter alia, name (product regulation), source, gunpowder, sexual form, identification, inspection, leachate, content determination, sexual taste and fate, function and management, artillery use, usage and quantity, attention, storage, intake standards, etc. The content of the income standard body shall be described on a case-by-case basis。
1. The name of the chinese medicine tablet shall reflect the chinese medicine, respect culture, inherit tradition and be named in accordance with the relevant provisions of the principles for the naming of drugs in china. Specific examples are: clean, cut tablets of biotoxic drugs, with the addition of the word “live” before the name of the drug, such as grass; fresh tablets, with the word “fresh” before the name of the drug, such as mint; chinese tablets, such as those made of fried, steam, scavenging, etc., with the name of the drug made in front of the tablet, such as plaster; or, if a gun is made of a variety of gunworks, named in the latter form, such as barbean frost; chinese tablets made with the aided cannon, such as beads; beverages, such as beverages, with the name of the beverage mixed with the cannon process, the name of the beverage is used as “subsilient + cannons” with the name of the platinum; and, if there is a variety of artillery processes, the termed in brackets, such as precorted yellow (cleaning) and precortuous yellow (drined wine stew)。
2. Sources include the chinese name of the primary source, i. E. The original plant, the latin name, the part of the drug use, the harvest season, the place of manufacture and the traditional name of the drug, among others; mineral medicines include the type, ethnicity, ore name or rock name of the mineral, the main ingredient and the place of production。
3. Artillery shall describe the key steps and parameters of the artillery process (e. G. Temperature, time, use of auxiliary materials, etc.) and the judgement indicators for meeting the requirements of the artillery。
4. Characteristics describe the main characteristics by the actual form of the drinking tablet。

5. Identification, including experience, micro-identification, physico-identification, thin layer identification, feature mapping, etc. The identification test shall be exclusive and durable。
If thin chromatography is used, the choice of standard substances and the preparation of their solutions, the preparation of the solution for the test, the sample, the plate, the extruder, the conditions (temperature, relative humidity, saturation time, etc.), the inspection method, etc. Should be examined。
In the case of liquid chromatography and gas chromatography, systematic applicability tests should be conducted to examine chromatography conditions such as chromatography columns, flow phase, current speed, column temperature, etc。
6. Inspections generally include the examination of toxicity limits (meaning drinking tablets with toxic ingredients), general impurities (water, total ash, acid insoluble ash, etc.), hazardous substances (heavy metals, arsenic salt, pesticide residues, sulphur dioxide residues, fungi toxins, microbiological limits, etc.), the methods and limits of which should be based on the requirements of the chinese pharmacy。
The leachate shall be subject to the relevant requirements of the chinese pharmacy and shall conduct necessary tests of the leachate, including solvents, leaching methods, etc。
8. The method by which the chinese pharmacy or provincial standard raw medicines are collected should be preferred. As far as possible, the determination of the content of a single ingredient or components above 2 ppm is generally not considered; as appropriate, the determination of the content of the active category components, such as total aqualone, total bioalkali, total soap, etc., may also be used; volatilizer content may be measured for volatile oil components。
The determination shall be validated in accordance with the relevant requirements of the chinese pharmacy. The content limits should be established with sufficient evidence and data accumulation。
9. The content of sexual tastes and attribution, function and management, usage and quantity, artillery use, etc., shall be regulated in the standard body and the basis shall be stated in the drafting notes。
(iv) stability tests
In accordance with the relevant technical guidelines issued by the directorate-general for the supervision of food and drug products, the conditions of storage of the product and its duration are determined through accelerated or long-term test visits。
(v) non-clinical safety evaluation studies

This is done in accordance with the relevant technical guidelines issued by the national directorate of food and drug control。
(vi) other
Direct oral tablets should establish a microbiological limit check and methodological validation as required by the chinese pharmacy。
Basic requirements for information to be requested
(i) description of standard technical information on chinese tablets
(1) draft standard for chinese tablets and drafting instructions. The drafting notes should fully reflect the entire process of the study (thesis, sample collection, research methodology, research results, etc.) and the necessary data, mapping, colour photographs, references, etc。
(2) the application submission of at least three sample self-inspection reports shall be in the same number as the sample to be sent to the sichuan food and drug laboratory for standard review。
(3) “sex and fate, function and principal, usage and quantity” of the drinking tablet should provide documentation or research on the effectiveness of clinical applications。
(4) the request for review by the standard-drafting unit itself shall also submit a request for approval of the chinese tablet standard. The applicant shall apply and provide a copy of the certificate of qualification, such as a unified social credit code, a business licence, a licence to manufacture medicines, a licence to practise medical institutions, etc. A description of the patent and the tenure status of the application for approval should be provided and a statement of commitment not constituting a tort should be submitted。
(ii) information format requirements
The paper is standardized in the international standard a4-type specifications, and the font is a chinese-speaking song, not less than a four-sign code, and each information should be covered and individually bound. The cover covers the name of the information project, the applicant (search), the date of application, the name of the test unit (search), the address, the telephone contact, the name of the person responsible for the test (signature), the date of commencement and termination of the test, the place of deposit of the original data, the name of the contact person, the telephone (with cell phone) etc。