Xinhua news agency, beijing, 10 december: promoting innovative medicines and medical devices with efficient and strict regulation - - visit to the party secretary and director of the national drug control authority, riley
Xinhua news agency journalist zhao wenjun, da xiaohe
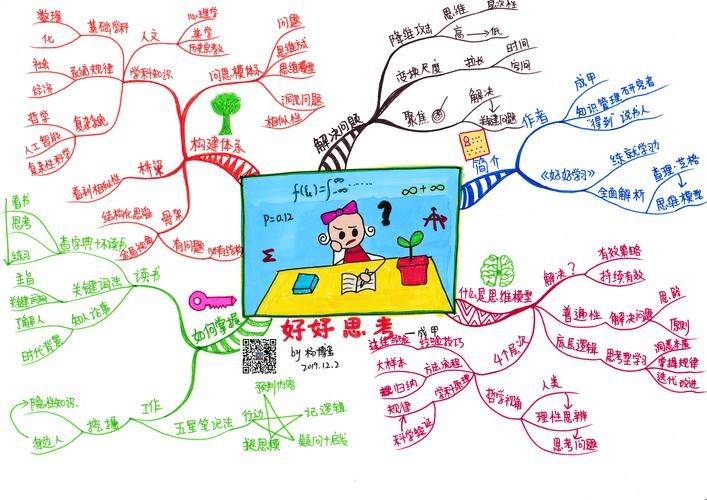
Medicines are a special commodity for the treatment of people in need, and ensuring their safety and security is a serious political issue, a basic human problem, a major economic problem and a critical technical problem. The recommendations of the central committee of the communist party for the formulation of the fifteenth five-year plan for the economic and social development of the country (hereinafter referred to as " the recommendations " ), which were considered at the twenty-fourth plenary session of the party, set out clear requirements for strengthening drug security and supporting innovative medicines and the development of medical equipment。
What has been the success of our pharmaceutical regulatory industry since the fourteenth five-year review? How can drug security be effectively maintained during the “155” period? How can research and development innovations in support of pharmaceutical enterprises be encouraged to facilitate the achievement of a high level of safety in medicines and a good development interaction in the pharmaceutical industry? Xinhua journalist interviewed the party secretary and director of the national drug regulatory authority, lee li。
Promoting solid steps in the building of a pharmaceutical power
Question: what have been the results of our pharmaceutical regulatory industry since 1455
Since 1405, the national drug regulatory system has adhered to the “four most stringent” requirements set out by general secretary xi, which have led to the deepening of drug regulatory reform, the establishment of a comprehensive drug safety floor, support for high-quality industrial development, the effective guarantee of overall stability in drug security and the promotion of firm steps towards the establishment of a pharmaceutical power。
• continued strengthening of regulations to guarantee the quality of medicines. Specialized anti-drug safety management and drug safety enhancement activities have been carried out to strengthen the monitoring of selected products, vaccines, blood products, implants, child cosmetics, etc., and to promote a chain-wide regulatory system covering all aspects of research and development, production and circulation. Each year, more than 200,000 batches of medicines are examined, more than 20,000 batches of medical equipment and 20,000 batches of cosmetics. The overall national drug-testing rate rose from 97. 8 per cent to 99. 4 per cent during the “135” period, and the drug safety situation remained stable over the long term。

• continuing deepening reforms to promote industrial innovation. (b) to develop a series of measures to improve the overall regulatory reform of pharmaceutical medical devices, and to develop the national drug control agency's opinion on deepening the regulatory reform of cosmetics for quality development in industry, in support of technological innovations in cosmetic raw materials. Since the fourteenth five-year plan, 220 innovative medicines and 282 innovative medical devices have been approved for listing, up from 6. 2 times and 3. 1 times respectively during the period of the thirteenth five-year plan. 415 children's medicines and 155 rare medicines have been approved for listing, 28 have been approved for listing, 344 new materials for registration and publication of cosmetics have been approved, and the pharmaceutical industry is gaining momentum。
• continuous upgrading of capacity to strengthen the regulatory base of medicines. (b) to promote the revision of the regulations for the implementation of the drug control act and the act on the management of medical equipment, among other things, and to establish a relatively well-developed legal regime for the regulation of pharmaceuticals. With the promulgation of the 2025 edition of the pharmacological code of the people's republic of china, the promulgation of 69 national regulations on tablets and tablets, 342 national standards for the formulation of chinese medicines and 481 standards for the medical equipment industry, the consistency of our medical equipment standards with international standards has been raised from year to year. Considerable progress has been made in the development of information on drug regulation, in the implementation of china's scientific plan of action for drug regulation, in the construction of a national priority laboratory for drug control science and in the implementation of 59 priority projects for regulatory science. The regulatory body has been fully strengthened by restructuring the national vaccine inspection centre, the special medicine inspection centre, the establishment of seven review and inspection sub-centres in areas where the pharmaceutical industry is concentrated, and capacity-building in drug regulation has entered the fast track。
Promote the transition of drug safety management from “passive defence” to “active control”
What measures have been taken to strengthen the regulation of drug safety during the “155” period
Response: drug security is an important component of public security. The drug regulatory authority will always take the most fundamental role in ensuring the safety of medicines, conducting thorough risk mapping, placing a high level of responsibility on the ground, and effectively maintaining the security and stability of medicines。
:: promoting improved drug safety liability systems. To improve overall preparedness to mitigate major risks and promote the transition from “passive defence” to “active control” for safe drug management. Optimizing cross-sectoral synergy mechanisms, ensuring that enterprises improve quality management systems, promoting the matching of local regulatory capacities with regional industrial development, and establishing clear, coordinated and efficient safety lines for high-quality development。
Strengthen chain-wide dynamic security controls. To take full advantage of the technical expertise of drug regulators, focusing on such priority areas as clinical trials, commissioning of production, network sales, to respond positively to new challenges such as product innovation, technological innovation and business innovation, to improve regulatory mechanisms covering research and development, production, operation and use of a full life cycle, to improve the regulatory and integrated inspection system, and to continuously enhance forward-looking, sensitive, predictable and proactive drug regulation。
Strictly combat violations. (b) regulate administrative inspection practices, strengthen supervision of the whole administrative enforcement process and regularity, and improve the overall normative level of administrative enforcement. To improve cross-regional cross-cutting mechanisms for the investigation of cases, to strengthen tracking, to implement the “punishment-to-person” requirement, to strengthen the chain of execution and discipline, and to promote joint inter-agency efforts to combat crime. Strengthen legal literacy education, explore new ways of alerting businesses to “speech” education, and raise awareness of compliance and rule of law in pharmaceutical enterprises。
Sustained support to enterprises to strengthen innovative drug development and development
How will the “fifty-five” period support the development of innovative medicines and medical devices
Response: innovative medicines and medical devices have high clinical value and technological content, which are key to driving our pharmaceutical industry from large to strong and are the basis for quality assurance of people's lives and health. Drug regulators will insist on an effective market and a combination of government, respect the innovative dominance of pharmaceutical enterprises, support enterprises in strengthening innovative drug development and development schemes, and help them transform new technologies into new quality productivity。
The first is to maintain policy direction and stimulate industrial innovation. Further expand the pilot results of reforms such as the approval of clinical trial reviews, the approval process for supplementary application reviews, and the production of segments, introducing “early intervention, one-size-fits-all, all-round guidance, joint research” on priority varieties, and placing the review resources fully in the direction of “new global” original products and nationally produced alternatives to “crawling” technologies, with a view to regulating policies to optimize the high-quality development of the pharmaceutical industry. To accelerate participation in the international drug inspection cooperation programme (pic/s), to initiate the evaluation of the world health organization (who) listing agency, to support the export trade in pharmaceutical medical devices and cosmetics, and to support open cooperation with the pharmaceutical industry。
The second is to insist on efficiency gains and accelerate the marketing of innovative medicines. Implementation of the national drug standards improvement programme, strengthening of technological advances in cell therapy products, radiopharmaceuticals, artificial intelligence products, and improvement of the technical principles and product standards for the registration of relevant products, with a view to optimizing the role of standards in the development of innovation. Optimization of the registration examination process, reduction of the number of registered samples tested, compression of the test cycle, reduction of the cost of the enterprise and reduction of the marketing cycle of the drug. To accelerate the improvement of the intellectual property protection system, such as the protection of experimental data on medicines, and to speed up the transformation of the original research results of pharmaceutical medical devices to enhance the core competitiveness of our pharmaceutical industry. (b) to improve the level of intellectual regulation, promote the regulation of artificial intelligence plus medicines, explore the implementation of artificial intelligence-assisted reviews, and promote scientific and technological empowerment。
The third is to maintain positive innovation and promote innovation in chinese medicine. The traditional advantages of chinese medicine are integrated into the requirements of modern drug research and development, and china's traditional chinese drug control system, which is consistent with china's characteristics, and the world's leading drug control system, is being improved. To promote the upgrading of traditional chinese medicine manufacturing, encourage support for the use of new technologies, techniques and formulations, and promote the conversion of medical preparations, older chinese medicine and national medicine into new chinese medicine。
Making drug regulatory reforms work for more patients
Question: how can the results of regulatory reform better reach the population during the “155” period

The recommendation clearly states that the principle of the primacy of the people must be adhered to. Drug regulation is closely linked to people's lives, and during the “155” period, drug regulation will always be based on increasing the people's sense of access, well-being, security and safety as a fundamental starting point and anchor for drug regulation, making the development gains of reform more and more equitable for all, and continuing to meet the people's aspirations for a better life and the need to protect their health。
Strengthening public awareness and accelerating the demand for clinical drugs -
(c) make full use of the accelerated access to breakthrough treatment drugs, conditional approvals, priority review approvals, special clearances, etc., to speed up the clinical approval of new medicines, rare diseases, children's medicines, etc. That are urgently needed outside the country, and to reduce the distance between laboratories and beds so that innovations can benefit people more quickly。
Consistent with standards and procedures, there has been a steady expansion of the evaluation of generic drug quality and efficacy consistency to reach more patients with affordable and good quality medicines. (b) actively promote the ageing of drug manuals and the pilot reform of accessibility, encouraging the provision of ageing and accessible versions of drug manuals, so that regulatory effectiveness is truly translated into satisfaction among the population。
Strengthening sectoral linkages for synergistic governance and development -
To strengthen coordination with the health, health and safety sectors, to strengthen clinical trial control, to continuously improve the system of drug retroactivity, to carry out a “clean-up” of drug operations, to develop inspection tools and techniques to adapt to new models of drug operations, to strengthen the monitoring and evaluation of adverse effects, and to ensure that medicines are safe and effective, so that people have access to medical care and medicines。
Strengthening multi-party cooperation and strengthening social governance in all its aspects —
Increased policy interpretation, presentation of results and awareness-raising for the general public, in a variety of forms and forms of broad-based community-wide drug regulation and regulation, high-quality development in the pharmaceutical industry and dissemination of information on safe medicines, continuous improvement of the population's scientific literacy of medicines, and creation of a sound public opinion environment that encourages support for the development and application of innovative pharmaceutical products. The main responsibility of third-party platform enterprises is secured, mutual care is maintained, and efficient mechanisms for risk prevention and control are put in place。