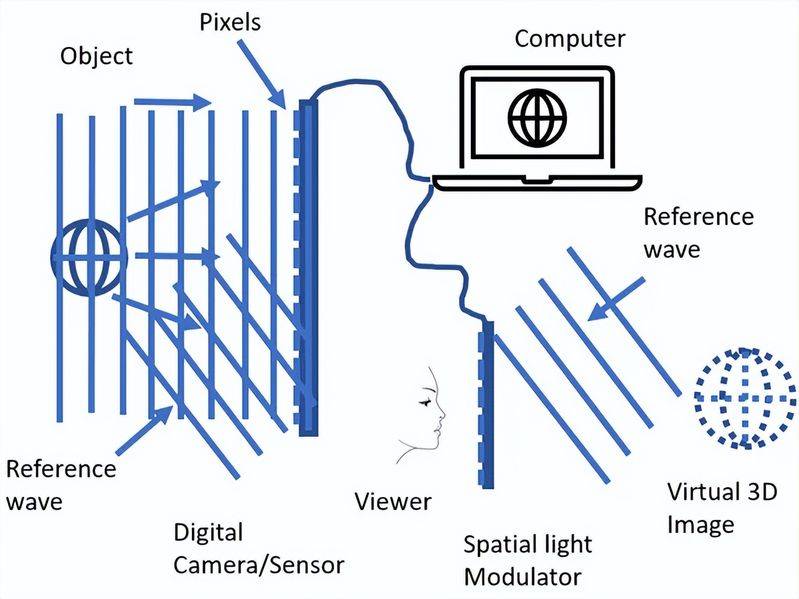
The warm baby is easy to carry and the heat is persistent. Thus, as an easy, safe, close-to-heat device, “heatbaby” has become a fashion appliance for international heating and cooling. So, what's its structural composition and the rationale it uses? Let's find out

Warm baby fever
Warm babies get hot because of iron's "oxidation"。
We know that iron blocks are very slow to oxidize under natural conditions, but if they are replaced with iron powder, the area of exposure to iron and oxygen is increased indefinitely, and water, salt and activated carbon form “original batteries”, which, with the help of a catalyst, will become relatively easy to oxidate to sub-iron, tritoxide and tritoxide. In general, the more stable the substance, the lower the energy; in turn, the more unstable the energy is. During iron oxidation, iron powder, as well as the intermediate products, iron oxidation and tritoxide, are unstable substances and further react to produce more stable iron tritoxide. So a lot of heat will continue to be released during the iron powder oxidation。
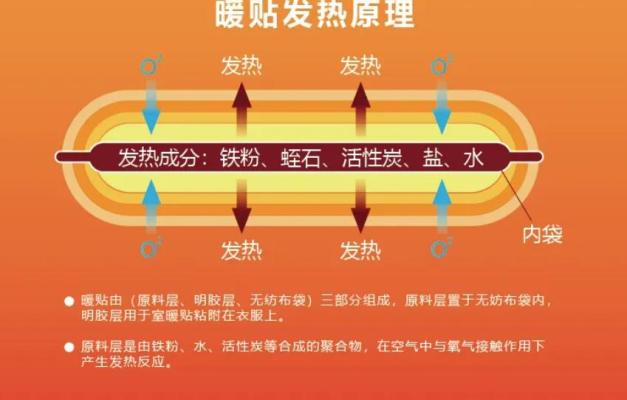
Detailed reaction component
Iron powder: oxygen in the air reacts to iron powder, which releases heat when it is corroded. These heats bring iron powder to about 68°c。
Small, porous bags: open plastic bags, air penetrates into a small, warm baby bag, where the chemical reaction to heat begins. The more small holes mean more air, so there are more small holes for warming a baby than normal。
Activated carbon: the role of activated carbon is to absorb the water in the air and ensure that the oxidation process is carried out smoothly. It also has thermal function, so it spreads the heat evenly。
Sodium chloride: in warm babies, sodium chloride (salt) mixed with water is a catalyst for iron powder corrosion。
Selenium: this water-bearing magnesium silicate will swell during heating. It's light, it's strong, it doesn't react chemically in warm babies, it doesn't smell, so it's a fireproof supermaterial. Neptunite is insulation and is used in some construction materials. Together with activated carbon, it spreads iron powder and avoids the heating of fillings in warm babies。
Source: cope shandong, cope for one minute